- Home
- About the Journal
- Peer Review
- Editorial Board
- For Authors
- Reviewer Recognition
- Archive
- Contact
- Impressum
- EWG e.V.
DOI 10.35630/2022/12/psy.ro.18
![]() ioana.silistraru@icloud.com, anamaria.ciubara@ugal.ro
ioana.silistraru@icloud.com, anamaria.ciubara@ugal.ro
Both pathologies—obesity and depression—have high prevalence rates and have serious negative effects on the public's health. In recent meta-analyses, clinical trials, and epidemiological studies, they have been observed in people of all races. Both obesity and major depression are risk factors related to one another. In this paper, we suggest an overview of the two interconnected biological processes, including genetic influences and changes to the systems in charge of energy synthesis and consumption (hypothalamic-pituitary-adrenal axis, and inflammation, neuroendocrine regulators, and gut microbiota). Additionally, we look into how people perceive their bodies and social stigma, as well as the potential benefits of physical activity and weight-loss surgery on comorbid conditions and quality of life.
Keywords: obesity, depression, bariatric surgery, gut microbiota, inflammation.
Being among the most significant issues with regard to global public health with increasing morbidity and mortality, obesity has recently attracted a lot of attention from the scientific community. The trend of rising life expectancy could be reversed as a result, according to predictions (Olshansky et al., 2005). Obesity is influenced by the intricate relationship between environmental factors, human behavior, and genetic predisposition. It is the biggest risk factor for many chronic illnesses, such as type 2 diabetes mellitus (T2DM), cardiovascular disease, pulmonary disease, renal disease, and various cancers. The World Health Organization claims that 13% of the world's population, or more than 1.9 billion adults, were obese in 2016.
Depression and obesity are common pathologies that tend to associate with individuals. The relationship was found to be bidirectional. There is evidence to suggest that they will become more common over the following years. Additionally, it is a financial burden due to the high cost of healthcare. Therefore, obesity and depression demand the attention of researchers and the scientific community (Vos et al., 2012).
Multiple studies prove that most of the time, obesity is associated with anxiety and depression (Brumpton et al., 2013). A U-shaped correlation between weight and depression was found in a cross-sectional survey carried out by Carey et al. in 2014 using data collected from 3,361 general practice patients. Up to 24% more underweight patients and up to 23% more obese people were found to have depressive symptoms on average. Only 11% and 12% of respondents reported finding these symptoms in normal-weight and overweight patients, respectively.
Trying to determine "Which came first, depression or obesity?" recent meta-analyses, clinical trials, and the majority of epidemiological studies all acknowledge a reciprocal relationship between the two (Blaine, 2008; Jantaratnotai et al., 2017; Luppino et al., 2010; Mannan, 2016). Obesity and mood disorders are linked risk factors. In cases of extreme obesity, the relationship appeared to be greater. (BMI ≥ 40 kg/m2). The heterogeneity of depression may cause some variability in association with obesity. Melancholic and atypical depression differ in symptoms. Loss of appetite and weight, impaired cognitive abilities, insomnia, anhedonia, and non-reactive mood are specific for the former, while lethargy, fatigue, sleepiness, increased appetite, and weight gain characterize the latter (Gold, 2015).
Another important factor that is frequently disregarded but may result in weight gain is simultaneous antidepressant therapy.
Table 1. Psychiatric medications and changes in body weight (Allison et al., 1999)

It has been demonstrated that hereditary variables can influence the reciprocal association between depression and obesity. Additionally, it affects the neuroendocrine regulators of energy metabolism, such as leptin and insulin, and the microbiome, as well as the homeostatic adjustment systems (HPA axis, immuno-inflammatory activation, and the neuroendocrine regulators of energy metabolism). Additionally, it results in changes to the brain circuitries that integrate homeostatic and mood regulatory responses (Milaneschi et al., 2019).
Recent literature (Robinson et al., 2017; Sullivan et al., 2000) proved that genetic factors influence obesity and depression in the same way and that there is approximately 40% heritability for both major depressive disorder (MDD) and body mass index (BMI). Through genome-wide association analyses, more than 200 loci related to BMI, obesity status, and fat distribution were found (Pigeyre et al., 2016). According to a 2015 study, the hypothalamus and pituitary gland, which regulate mood and energy homeostasis, exhibit strong expression of the genes close to BMI-associated loci, according to Finucane et al. (2015) more than 50 genetic loci connected to depression phenotypes have been found in terms of MDD genetics. The genes with the strongest signals were those that had previously been linked to severe early-onset obesity and were located close to one another or overlapped (Wheeler & Pierce, 2013). The majority of shared mechanisms also frequently reflect this shared genetic foundation.
One of the most significant findings in biological psychiatry is that when the hypothalamic-pituitary-adrenal (HPA) axis is overactive, cortisol is released uncontrolled and unabatedly. Long-term cortisol overuse damages and depletes neurons in limbic regions that are sensitive to stress and linked to depression (Schmaal et al., 2016). The relationship between mood disorders and high cortisol levels is also apparent in Cushing's syndrome patients (CS). Pituitary or adrenal adenomas can result in endogenous hypercortisolism, and 50–80% of individuals with active illness had MDD. The emergence of depressive symptoms and their recovery after surgery provide compelling evidence for the association between high cortisol levels and MDD (Milaneschi et al., 2019). According to the same author, in almost half of the adult obese population, high long-term cortisol levels can be found. Even at a young age, HPA axis hyperactivation increases the risk of obesity 10-fold.
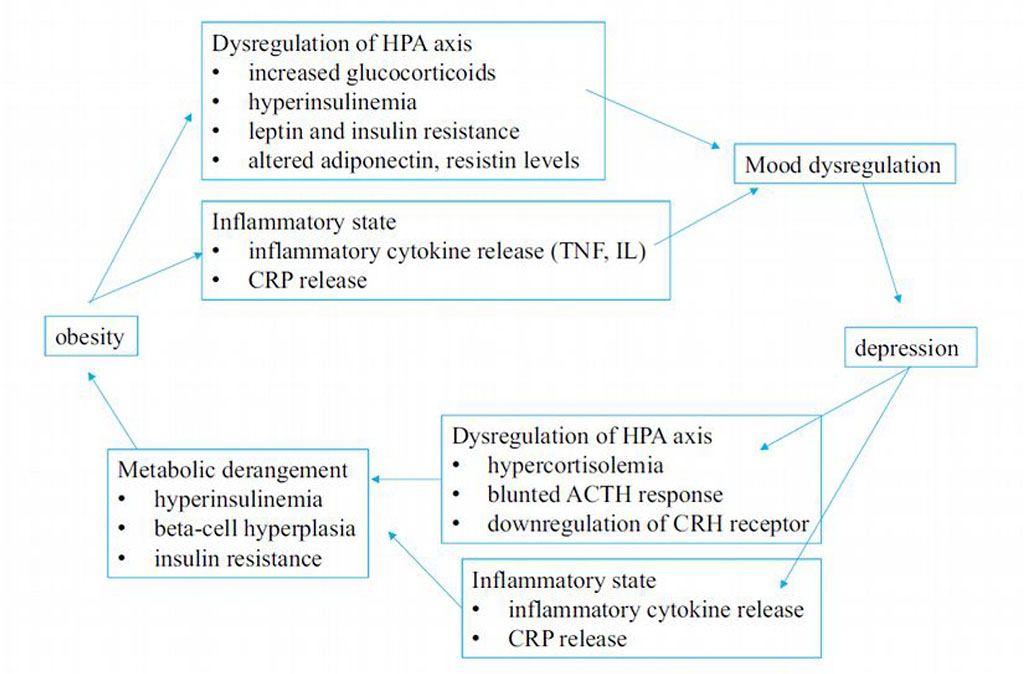
Figure 1. The bidirectional connection between
obesity and depression
Source: Authors'
own conception
Increased cravings for high-calorie foods, encouragement of adipogenesis and visceral fat tissue hypertrophy, suppression of thermogenesis, and a decrease in energy expenditure are the main factors that lead to obesity in patients with high cortisol levels. It thus tends to make them more prone to metabolic comorbidities and depression (Fardet & Fève, 2014).
Concerning cortisol release and metabolism, the functioning of the glucocorticoid receptor (GR) is disrupted by the chronic inflammation (proinflammatory cytokines) characterizing obesity. Additionally, dysfunction of the 5α-reductase enzyme necessary for glucocorticoid clearance increases the build-up of adipose tissue and the onset of MDD. 11-β-hydroxysteroid dehydrogenase is a protein that converts bioactive cortisol into inactive cortisone and vice versa (Mansour et al., 2016). Non-alcoholic fatty liver disease (NAFLD), which is linked to decreased activity of the enzymes in charge of cortisol clearance and regulation, is one of the comorbidities that frequently go in tandem with abdominal obesity.
Tumor necrosis factor (TNF-), interleukin (IL)-1, IL-6, and other pro-inflammatory cytokines are commonly produced in response to infections and malignancies. Anorexia and the wasting syndrome connected to these illnesses have been related to these cytokines. The mentioned cytokines cross the blood-brain barrier and influence neuronal pathways in the central nervous system to regulate eating behavior and energy balance (CNS) (Plata-Salaman, 1998). The complete evaluation of inflammatory markers in studies is scarce. High levels of IL-6 and C reactive protein have been found to be the most frequently associated findings with obesity and depressive symptoms (CRP). BMI was known to be involved in seven research studies on 9,421 people as the main driver of low-grade systemic inflammation. Four additional studies involving 16,837 participants emphasized the fact that high CRP levels continued to be linked to depressive symptoms even after BMI had been adjusted. This suggests that peripheral inflammation, even in the absence of obesity and overweight, can cause depressive symptoms (Ambrósio et al., 2018). Inflammation and adaptable behavioral changes only last a short while as part of the normal immune response. If the response is exaggerated or prolonged, several brain areas responsible for managing satiety, energy balance, cognition, and mood may be impacted. Inactivity, weight gain, and permanent metabolic changes can all result from this (Castanon et al., 2014; Markowitz et al., 2008).
The arcuate nucleus (ARC), a critical regulator of energy balance, is located in the mediobasal hypothalamus, close to the bottom of the third ventricle. Neurons in this region express a number of hormones that control appetite, including leptin, cortisol, oestrogen, progesterone, and growth hormone. The ARC can be considered of as a "metabolic sensor" as it receives and integrates metabolic and endocrine information regarding nutritional and energetic status from the periphery. (Williams et al., 2001).
The role of leptin is crucial. In both animal and human studies, it has been demonstrated that genetic deficiencies of either leptin or its receptor result in severe neuroendocrine and metabolic abnormalities, hyperphagia, and morbid obesity (Ahima et al., 2000). Despite having elevated levels of circulating leptin, the majority of obese patients associate forms of leptin resistance, a condition similar to insulin resistance in type 2 diabetes mellitus, which significantly reduces its anorexigenic effect. Central signaling mediated by leptin is also altered by obesity-related inflammation. Chen et al. concluded in a 2006 study that high levels of CRP can directly inhibit leptin binding to its receptors. Leptin treatment had antidepressant effects in behavioral testing on animal models, and it also corrected depressed behavior brought on by prolonged stress (Garza et al., 2012).
The insulin pathway, which controls glucose metabolism and eventually results in T2DM, is also altered and disrupted by obesity. Insulin resistance is the early stage condition in which peripheral tissues stop responding to increased insulin release from the pancreatic beta-cell. Again, these modifications appear to be brought on by high levels of circulating pro-inflammatory cytokines that reduce the receptor's capacity to spread the signal and trigger pancreatic β-cell apoptosis, according to Milaneski et al. The inhibition of memory and executive function, as well as neuronal damage in the medial prefrontal cortex and hippocampus, are related to insulin resistance and the modification of regional cerebral metabolism. This may suggest that it plays a role in some neuropsychiatric diseases including depression and dementia (Rasgon & McEwen, 2016). Furthermore, a bidirectional association between T2DM and MDD is suggested by several recent meta-analyses (Mezuk et al., 2008; Rotella & Mannucci, 2013).
Human health and its interdependent, strong relationship with the microorganisms that live in the gut have received more scientific attention in recent years. Over 100 trillion microorganisms (bacteria, archaea, yeasts, single-celled eukaryotes, parasites, and viruses) reside in the gastrointestinal (GI) tract and are involved in the majority of physiological functions, including digestion, homeostasis, and yeast development. Recent studies have emphasized how the gut microbiota affects the gut-brain axis and may play a part in conditions that affect the central nervous system and neuropsychiatric disorders (Cryan & Dinan, 2012; Foster & McVey, 2013). This indicates a two-way communication between the CNS and the gut microbiota. Autonomic, neuroendocrine, and immune pathways are involved, though the exact mechanism of action is not yet fully understood.
A third of the microbiota in our gut is shared by all humans. The remaining two-thirds are unique to each person. Although it is challenging to define a healthy microbiota due to its distinctive characteristics, it is generally accepted that it includes species diversity and community stability. Bacteria from the phyla Bacteroidetes and Firmicutes are the two most prevalent in humans (the latter producing more harvestable energy). An altered ratio between the two phyla, favoring Firmicutes, is a hallmark of obesity. This ratio was also linked to signs of local inflammation, which may make the gut more permeable to bacteria and start a systemic inflammation (Cani et al., 2008).
Studies on both humans and animals demonstrate that the microbiota-gut-brain axis and the inflammatory response are influenced by anxiety and depression, two conditions that affect millions of people worldwide, as a result of an abnormal ratio between the two major bacterial phyla (Rieder et al., 2017). Preliminary research on microbiota transplantation from patients with MDD-induced depression-like behavior in rats (Kelly et al., 2016; Zeng et al., 2016).
The reduction of time spent with physical activity, sedentarism, western diets, and the consumption of high-caloric foods are the main drivers of obesity over the past decades. These risk factors are also common in persons with mood disorders, making them more likely to engage in smoking, excessive alcohol consumption, poor nutrition, and sleep hygiene, behaviors associated with depression and obesity (Ciubara et al., 2018; Milaneschi et al., 2018; Silistraru et al., 2022).
The literature states that psychological and lifestyle factors come second in the etiology of obesity after genetic factors. Still, while the latter is relatively historically unchanged, lifestyle factors change over generations, and obesity in modern society is increasing (Lissner, 1997). In addition, the rising rates of obesity correlated, according to Lissner, to social-environmental factors, with increased food intake and lowered energy expenditure in modern society. However, unpublished data from a Swedish study by Lissner and Bengtsson focused on the correlation between obesity and education in women, showing that lower rates of obesity appear in more educated women in the late sixties and early nineties (Lissner, 1997). The findings impact the elaboration of public health policies and education in general.
An essential component of personal identity is body image. It can be defined as the figure one has on their anthropometric measurements, shapes, and body contours. Satisfaction with one’s body shape or specific body parts is related to these factors (Gardner, 1996). Both obesity and depression proved to influence body image perception. This can promote or aggravate specific comorbidities or eating disorders (Paans et al., 2018). Social stigma is represented by the discrimination against or the disapproval of certain persons based on perceivable social characteristics that distinguish them from other members of society (Luca et al., 2020). Obesity-related stigma is omnipresent in contemporary societies. Overweight people are perceived as lazy, unintelligent, unpleasant, and lacking willpower (De Brun et al., 2014). Teasing and stigmatization, however, do not motivate weight loss. Instead, it can contribute to the development of distress, depression, low self-esteem, and disordered eating behaviors when combined with factors like gender, age, level of obesity, parental criticism, and binge eating (Radulescu et al., 2020). Multiple studies positively associate experiencing social stigma with developing binge eating or emotional eating behaviors and depressive symptoms (Ashmore et al., 2008; Farrow & Tarrant, 2009; Friedman et al., 2008; Savoy, 2010; Wott & Carels, 2010; Wu & Liu, 2015). It seems that women idealize a lean body type and perceive their body as larger than reality, as opposed to men who idealize a larger, more muscular body and perceive themselves as underweight (Silva et al., 2019).
The experience of social and educational discrimination is measured in the obese population (Sarwer & Polonsky, 2016) with occurrence in several settings. Overweight individuals are less likely to graduate from high school, enter a marriage, or access employment compared to the normal-weight population (Friedman et al., 2008). The measured levels of discrimination are likely to increase for patients suffering from severe obesity, according to the literature. While researching the age segmentation of patients suffering from stigma manifestation and consequential discrimination, data extracted from literature reviews prove that negative perception starts at an early age with overweight children (Burlea et al., 2012; Harriger & Thompson, 2012). The literature shows preschool children’s preference for slimmer body composition (Harriger & Thompson, 2012) compared to normal or overweight figures. Teenagers and young adults are at greater risk of discrimination as a result, which can expose them to particular health and mental issues such as body image problems, eating disorders, impaired self-esteem, and psychological effects like severe depression and suicidal thoughts (Harriger & Thompson, 2012, p. 5; Sarwer & Polonsky, 2016).
Dankel et al. (2016) negatively correlated the duration of overweight/ obese conditions and lack of physical activities with quality of life levels. Different types of physical training can be a complementary and effective strategy for improving quality of life and emotional health. They can also improve most comorbidities associated with obesity and depression. It can also improve motivation, self-esteem, mood, and psychological state (Vancini et al., 2017).
Bariatric surgery consists of several surgical procedures performed to facilitate weight loss. The most popular procedures performed globally are sleeve gastrectomy (SG) and laparoscopic Roux-en-Y gastric bypass (RYGB). In comparison to dietary recommendations, exercise, lifestyle changes, and medication, there is already a sizable body of evidence that supports bariatric surgery as the most appropriate method for important and sustained weight loss, with significant amelioration of comorbidities or even remission (such is the case of T2DM). This growing evidence led some obesity experts to see the surgical approach as the solution to the looming obesity epidemic (Jumbe et al., 2017). Specific literature identifies a higher prevalence of mood disorders, anxiety, alcohol use, low self-esteem, personality, and eating behavior disorders in bariatric surgery candidates than in the control group or obese patients who do not seek a surgical procedure (Pull, 2010). Despite significant weight loss and improvements in physical health, there is currently little information available regarding long-term psychological health outcomes following bariatric surgery. De Zwaan and colleagues (2011) looked at anxiety and depressive disorders in 107 bariatric patients who were morbidly obese before and 6, 12, 24, and 36 months later. He found that the prevalence of anxiety and depressive symptoms decreased significantly after the surgery, but these patients lost less weight than predicted.
Obesity and depressive disorder are common pathologies with overlapping etiopathology. They both have a hereditary component, and their association may produce adverse health outcomes. Treatment options should always begin with prevention and pre-emption. There is evidence to back up the claim that bariatric surgery is the most effective method of treating obesity and its associated comorbidities, however patients should be carefully evaluated and chosen.
For various psychiatric disorders like anxiety and depression, a more novel approach might soon be accessible. Early preclinical research indicates that changing and modulating the composition of the gut microbiota may have effects on behavioral health.
Both obesity and depression may respond well to treatment approaches that focus on the shared mechanisms mentioned above. Targeting inflammation might be an encouraging lead. Anti-inflammatory drugs reduced patients' depressive symptoms, according to a meta-analysis of 14 placebo-controlled trials (Köhler et al., 2014). Additionally, effective treatment plans for one condition might be expanded upon and put to the test for the other.
This work was supported by a project funded by the RO-MD Cross-Border Program, Priority 4.1 - "Support to the development of health services and access to health", project code: 1HARD/4.1/93