- Home
- About the Journal
- Peer Review
- Editorial Board
- For Authors
- Reviewer Recognition
- Archive
- Contact
- Impressum
- EWG e.V.
DOI 10.35630/2022/12/psy.ro.11
Polyradiculoneuritis or Guillan Barré’s Syndrome is the most common cause of acute and subacute generalized paralysis, which is likely to occur at any age. We present the case of a 17-year-old adolescent who was admitted to our clinic for swallowing and phonation disorders, evolving into acute respiratory insufficiency (which required orotracheal intubation and ventilatory support), hypotonia in the upper and lower limbs, and globally-decreased osteotendinous reflexes, abolished in the upper limbs. The thoraco-abdominal X-ray and the chest CT revealed lower respiratory infection, and the neurological clinical examination and the lumbar puncture with albino-cytological dissociation raised the suspicion of polyradiculoneuritis. Therapy with intravenous immunoglobulin resulted in the improvement of the motor deficiency, but without the possibility to detubate the patient. Following 2 sessions of plasmapheresis with 20% human albumin, significant improvement of the neurological deficiency was seen and spontaneous breathing was resumed, the patient regaining full muscle strength.
Keywords: polyradiculoneuritis, Guillan Barré’s Syndrome, intravenous immunoglobulins, plasmapheresis, swallowing disorders, pneumonia
Acute polyradiculoneuritis is an immune-mediated inflammatory polyneuropathy causing acute and diffuse demyelinating lesions in the nerves of the peripheral nervous system (Korinthenberg, 2013). In approximately 60% of cases, it occurs after a respiratory infection (most commonly caused by Mycoplasma pneumoniae) or after a gastrointestinal infection (Campylobacter jejuni, Helicobacter pylori), with neurological symptoms occurring after 1-3 weeks. It is a non-epidemic and non-seasonal disease, with a distribution women/man of 1.5/1. The earliest symptoms are paraesthesia of the lower limbs, after which the progressive, relatively symmetrical, ascending motor deficit is installed, affecting the intercostal muscles and the neck (Mullings et al., 2010). The clinical picture raises the suspicion of polyradiculoneuritis (also known as Guillan Barré’s Syndrome), and confirmation is made by examining cerebrospinal fluid and by needle electromyography. The specific therapy requires admission in intensive care unit for surveillance, because the ascending paralysis can rapidly develop into acute respiratory failure; without orotracheal intubation and ventilation support it leads to death. The therapy with the best results is the administration of intravenous immunoglobulin 0.4g/kg/day for 5 days. If this protocol is insufficient, plasmapheresis is another option (Bersano et al., 2006; Doets et al., 2020; Luo, 2020).
An adolescent, aged 17 years and 11 months, was admitted to our clinic for irritable dry cough, 39°C fever, weak voice, mixed dysphagia, loss of appetite, sialorrhea; these symptoms started a week ago. At home he was treated with antibiotics, according to the recommendations of the family doctor, but he cannot remember the name of the medicine. The patient came without a legal companion, and the anamnesis was performed with difficulty due to major dysphonia and dyslalia. The family medical history did not reveal significant pathological elements, and the personal history shows that he was vaccinated according to the national regulations and that he did not undergo any chronic therapies at home.
The clinical examination upon admission showed satisfactory general condition; the patient was of normal weight, aware, cooperative, time and space oriented, facies with dark circles, pale skin, loaded tongue, normal pulmonary sounds, weak voice, irritating cough and suprasternal retraction.
Laboratory tests were: leucocytosis (16.990/mm3) with neutrophilia (13.650/mm3), increased inflammatory markers (ESR-33mm/h, Fibrinogen-531 mg/dl), hyposideremia (Iron-20 µg/dl), hypocalcaemia (Ionic Calcium - 4 mg/dl) and the increase of the alkaline reserve (AR-33. 22 mmol/l).
Based on the clinical and paraclinical criteria, the diagnosis of acute supraglottic laryngitis was established and we began the therapy with Ceftriaxone, hydrocortisone hemi succinate and symptomatic medication.
During the second day of hospitalization, the patient's condition worsened, with marked sialorrhea and swallowing disorder, accentuated dyspnoea with polypnea, generalized retraction, tachycardia, oxygen saturation being 84% in atmospheric air and 89% with oxygen source. ENT examination was recommended, and the indirect laryngoscopy revealed the lack of oedema in the epiglottis and salivary stasis in large quantity in the pyriform sinuses, thus discarding the diagnosis of acute supraglottic laryngitis. The patient was admitted to Intensive Care and submitted to therapy with Meropenem and Vancomycin, facial mask oxygen therapy and symptomatic medication. The blood count showed an increase in leucocytosis and neutrophilia. Reactive protein C, initially within normal limits, increased to 130 mg/l.
On the third day, due to the progressive deterioration of the general condition, anxiety and suffocation under oxygen therapy, the patient was sedated and orotracheal intubation was applied, increasing saturation from 82% with oxygen source to 92% after intubation. During the manoeuvre the absence of the pharyngeal reflex was noted (following the direct stimulation of the pharynx there was no feeling of nausea or vomiting, nor the contraction of the pharyngeal wall muscles). Nasogastric tube, central venous catheter on the internal jugular vein, and urethro-bladder tube were applied.
A number of additional paraclinical examinations were recommended for the diagnosis: sample of tracheobronchial aspirate, nasal exudate, haemoculture, urine culture, coprocytogram and stool culture, but all these do not lead to a specific etiological factor. Ac VHC, Ag HBs, Ac anti Mycoplasma pneumoniae IgM, Ac anti Borrelia burgdorferi IgM, Ac anti Cytomegalovirus IgM, Anti-HIV 1+2 were negative. Front-rear thoracic X-ray reveals a medium-intensity dull structure, unclearly delimited, without the aerial bronchogram visible in the lower half of the left hemithorax, with moderate attraction effect of the lateral intercostal areas (figure 1).
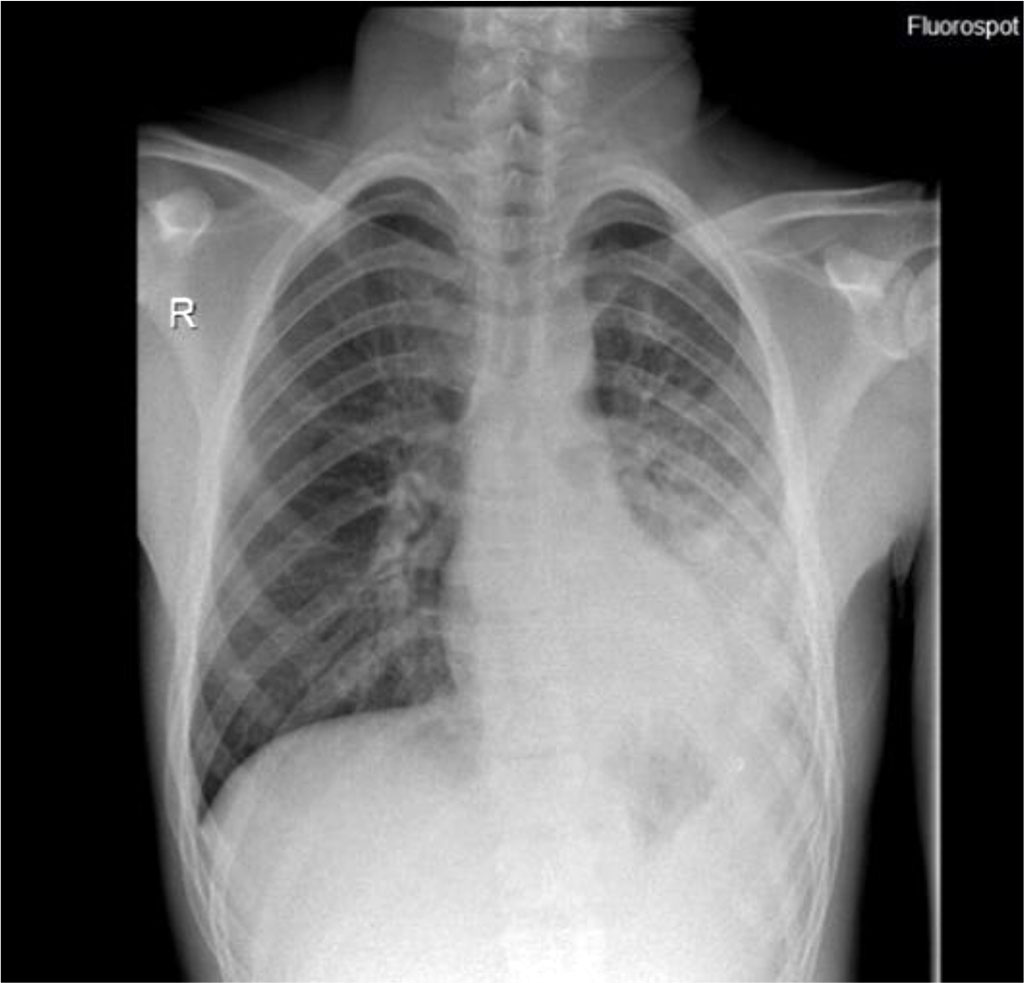
Figure 1. Thoraco-abdominal X-ray - dull structure unclearly delimited in the
left hemithorax.
Source: Authors' own work
CT examination revealed focal alveolar filling without aerial bronchogram, which contained small aerial inclusions, with normal contrast of the vascular structures, visible at the level of the lower left lobe, which moderately exerted a retractable effect on the fissure – atelectasis of the lower left lobe. Small amount of left pleural fluid in basal rear area, ~ 5mm (Figures 2, 3).
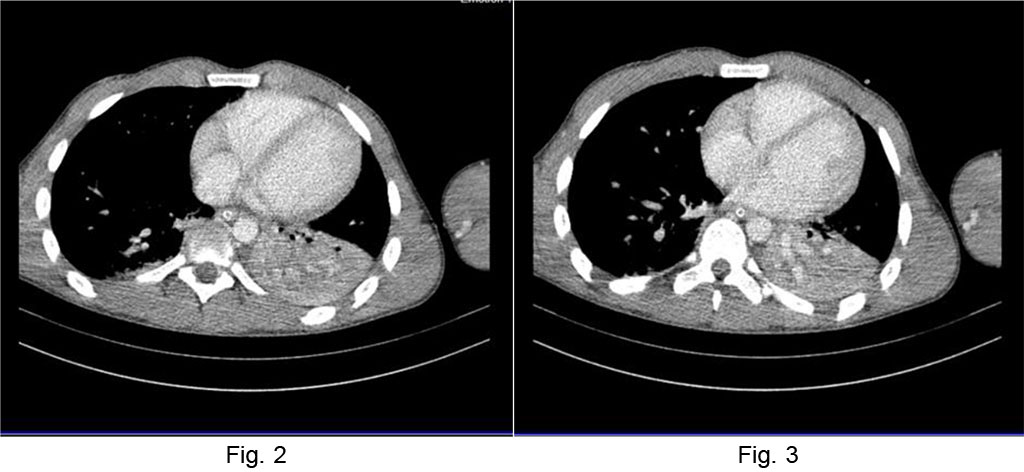
Fig. 2 Chest CT atelectasis
of the lower left lobe Fig.
3 Left
pleural fluid layer.
Source: Authors' own work
For a final diagnosis, the tuberculin intradermoreaction was performed; after 72 hours, a palpable dermal papilla appeared, uneven, with a diameter of approximately 15 mm. Tracheal aspirate was negative, with no mycobacterial DNA. The patient continued to receive Vancomycin and Meropenem, to which an anti-fungal was added. Significant amounts of purulent secretions were aspirated from the oro-tracheal intubation tube. After 5 days of antibiotic therapy it was decided to decrease the sedative dose and to detubate the patient, but it was found that the patient, although aware and cooperative, could not resume the spontaneous breaths as the cough reflexes were absent, and the limb muscles were hypotonic. The patient also had facial paresis. Lumbar puncture was performed, and the analysis of cerebrospinal fluid showed albumino-cytological dissociation.
Following the neurological clinical examination, we found the global decrease of the osteotendinous reflexes and the decrease of the muscular force predominantly in the upper limbs. The suspicion of polyradiculoneuritis was raised, and for the precision of the diagnostic the needle electromyography was recommended. The result suggested polyradiculoneuritis.
Intravenous human immunoglobulin treatment was initiated, 0. 4g/kg/day, for 5 days, with improvement of motor deficiency, but with the impossibility of detubation due to persistent respiratory failure. Subsequently, two sessions of plasmapheresis were performed, with 20% human albumin, resulting in significant improvement of the neurological deficit. The patient was detubated and spontaneous breathing, standing and walking were resumed. At 28 days after the admission the patient was discharged with a very good general condition, with the diagnosis of acute polyradiculoneuritis.
Polyradiculoneuritis is the most common cause of acute or subacute paralysis. From the etio-pathogenic point of view, primitive polyradiculoneuritis, which occurs in the absence of a premonitory disorder, along with secondary polyradiculoneuritis developing in a pathological context: infections (viral hepatitis B and C, cytomegalovirus, Epstein Barr’s virus, HIV, Mycoplasma and Campylobacter), vaccinations (rabies, oral polio and conjugated antimeningococci), surgery and blood transfusions (Van Doorn et al., 2008). The pathogenesis is partially known, the most plausible hypothesis being the occurrence of an autoimmune process. Acute demyelination blocks the transmission of nerve impulses favouring the onset of clinical symptoms. When the cause is cleared, the normal myelination process is resumed (Hauser & Josephson, 2013).
This case started with a respiratory infection in a patient with no significant personal history, and about 10 days after the onset, swallowing and phonation disorders caused the adolescent to come to the hospital.
Judging by the symptoms and the conclusions of clinical examination, several possible diagnostics were considered initially: aspiration of foreign bodies, retropharyngeal/peritonsillar abscess, acute supraglottic laryngitis. Although the patient had coughing and choking seizures, the diagnostic of aspiration of foreign bodies was discarded after anamnesis. The retropharyngeal/peritonsillar abscess occurs with sialorrhea, swallowing disorders and trismus, but it doesn’t affect the voice, so this diagnosis was also discarded. Dysphagia, sialorrhea, suprasternal retraction and weak voice were the main clinical manifestations that guided us towards the diagnosis of acute supraglottic laryngitis. Saliva allows effective chewing, swallowing and speaking. Also, saliva has antibacterial and antifungal properties, thus playing a protective role (Ignat et al., 2017).
The patient’s condition aggravated progressively. The biological samples showed no specific pathogens, and the lumbar puncture, after the sampling and analysis of the cerebrospinal fluid, supported the diagnostic of polyradiculoneuritis. Although the clinical picture was atypical, lumbar puncture and needle electromyography confirmed once more the diagnostic of Guillen Barré’s Syndrome. The therapy with immunoglobulins was not sufficient for the detubation of the patient, but after the two sessions of plasmapheresis, the patient resumed effective spontaneous breathing, standing and walking, being discharged with very good general condition.
The peculiarity of our case is represented precisely by the onset of polyradiculoneuritis, by affecting the bulbar cranial nerves, with swallowing and phonation disorders, being this a less common clinical picture. In the literature, the administration of immunoglobulins and plasmapheresis are considered to have relatively equal benefits (Mullings et al., 2010). The second peculiarity of the case is the aggressiveness of the disease, the evolution being progressively aggravating despite the administration of immunoglobulins, which required the addition of plasmapheresis. The pathology is on its natural healing course. Our patient has completely regained muscle strength, but some patients may be left with residual weakness. In about 7% of cases, children with polyradiculoneuritis may have a recurrence of the disease or it may be chronic and taking the form of chronic demyelinating inflammatory polyradiculoneuritis (Kliegman et al., 2007).
Also, cases of Guillan Barré’s Syndrome have been reported as having as a trigger factor the implantation of an allergenic bone graft in oro-maxillofacial surgery. A 55-year-old patient required restorative surgery for the completely edentulous and atrophic mandible and jaw. Bone autograft from the left iliac crest combined with lyophilized allografts were used to restore the thickness and height of the mandible and jaw, and postoperative evolution was favourable; however, after 7 days, the patient experienced episodes of muscle weakness and paralysis of the lower limbs, followed by respiratory insufficiency because the intercostal muscles were affected. After the specific therapy, the muscle deficit was improved, the patient was left with no residual paralysis or other neurological deficiencies (Cicciù et al., 2015; Vendemmia et al., 2019).
Polyradiculoneuritis or Guillan Barré’s Syndrome is a disorder of the nerve roots and peripheral nerves, with possible extension to the cranial nerves, due to an extensive inflammatory process. In most cases, with the help of appropriate therapy, the patients recover completely. The supportive treatment is the admission to the Intensive Care Unit, and the pathogenic treatment involves intravenous immunoglobulin and plasmapheresis.