- Home
- About the Journal
- Peer Review
- Editorial Board
- For Authors
- Reviewer Recognition
- Archive
- Contact
- Impressum
- EWG e.V.
Cite as: Archiv EuroMedica. 2025. 15; 4. DOI 10.35630/2025/15/4.001
Background: Annual influenza vaccination is recommended in many countries due to the rapid antigenic drift of circulating strains, particularly influenza A (H3N2). Evidence indicates that vaccine effectiveness may decline after repeated administrations, partly due to antigenic imprinting, although cellular immunity often remains stable or improves. Understanding the long-term immunological impact of repeated vaccination across age groups is essential for optimizing public health strategies.
Aim of the study: To evaluate the effect of repeated influenza vaccination on vaccine effectiveness and immunological outcomes in the general population.
Material and Methods: A systematic review was conducted in accordance with PRISMA 2020 guidelines. Studies published between January 2020 and March 2025 were identified through PubMed, Google Scholar, and ResearchGate using predefined search strings. Eligible studies reported on repeated or annual influenza vaccination in humans and measured humoral or cellular immune outcomes. Risk of bias and methodological quality were assessed using validated tools. Twenty-eight studies met the inclusion criteria.
Results: The studies varied in population characteristics, design, and vaccine type, with most using inactivated formulations. Immune responses were assessed mainly through antibody titers, seroconversion rates, and in some cases, cellular immunity markers. Findings were heterogeneous: 10 studies reported a negative effect of repeated vaccination, 11 showed a beneficial effect, and 7 found no significant impact. When reported, effect sizes were generally small to moderate. Statistical significance was inconsistent, and clinical relevance was frequently unclear. Observed effects were influenced by age, immune status, prior vaccination history, and vaccine formulation.
Conclusions: While repeated vaccination may attenuate humoral responses in some populations, particularly older adults, cellular immunity tends to remain stable or improve, contributing to sustained protection. Personalized strategies, such as high-dose or adjuvanted vaccines for the elderly, may help offset reduced antibody responses. Limitations of the current evidence include heterogeneity in study design, absence of standardized outcome definitions, inconsistent statistical reporting, and limited long-term follow-up. Future research should incorporate robust bias assessment, standardized immunological endpoints, and age-stratified analyses to clarify mechanisms and optimize vaccination strategies.
Keywords: influenza vaccine, repeated vaccination, annual vaccination, immune response, vaccine effectiveness, antigenic imprinting, humoral immunity, cellular immunity
Annual influenza vaccination is necessary due to the rapid antigenic drift of circulating strains, particularly influenza A (H3N2), which requires frequent reformulation of the vaccine. Immunity against homologous strains wanes over time, supporting the need for yearly immunization. In many countries, annual influenza vaccination is part of public health recommendations. However, evidence suggests that vaccine effectiveness may decrease after repeated administrations. Influenza remains a major health problem because of its direct clinical impact and its broader social and economic consequences, especially among the elderly, people with chronic diseases, and immunocompromised individuals [1,2].
Recent studies indicate that repeated vaccination may reduce antibody responses in some cases, particularly in older adults, partly due to immunological imprinting, also referred to as original antigenic sin. For example, Sullivan et al. [1] reported reduced vaccine effectiveness against A(H3N2) in individuals vaccinated in consecutive seasons, whereas Guiomar et al. [13] found that repeated vaccination maintained protective antibody levels even against mismatched strains. In contrast, cellular immunity appears more stable and may even improve with repeated vaccination, indicating a complex pattern of immune protection [3,4].
While some earlier reviews have addressed this topic, many have overlooked recent findings on age-related variability in immune responses. For example, Kitamura et al. [6] observed no significant change in antibody titers after repeated vaccination in elderly individuals, whereas Matsumoto et al. [11] reported maintained or improved vaccine effectiveness in young children receiving annual doses. Similarly, Fox et al. [2] demonstrated that enhanced vaccine formulations in older adults can extend antibody reactivity, although prior vaccination effects persist. In particular, there is a lack of comprehensive data on the long-term immunological effects of repeated vaccination across different age groups, on how prior vaccination history modifies both humoral and cellular immunity, and on whether specific vaccine formulations can mitigate potential reductions in effectiveness. The role of targeted vaccination strategies, including high-dose and adjuvanted formulations, has been insufficiently examined.
The present review focuses on studies published between January 2020 and March 2025, retrieved exclusively from PubMed, Google Scholar, and ResearchGate, and limited to articles written in English. These restrictions, along with the absence of a formal risk of bias meta-analysis, should be considered when interpreting the findings. The search was guided by predefined key terms including “influenza vaccine”, “repeated vaccination”, “annual vaccination”, “immune response”, “immunoprotection”, and “antibody titer”.
This review aims to address existing knowledge gaps and to provide evidence-based insights to inform clinical decision-making and public health immunization policies. Understanding the long-term effects of repeated vaccination is essential for optimizing strategies and improving protection for populations at highest risk of severe influenza outcomes. In this context, a systematic review of recent studies can clarify the magnitude and direction of the impact of repeated vaccination on different components of the immune response and on overall vaccine effectiveness.
A systematic literature review was conducted in accordance with the PRISMA 2020 guidelines. Three databases - PubMed, Google Scholar, and ResearchGate - were searched for relevant peer-reviewed studies published between January 2020 and March 2025. The review protocol was not registered in PROSPERO or other registries, which is acknowledged as a methodological limitation.
Studies were included if they:
Exclusion criteria:
Searches were performed in PubMed, Google Scholar, and ResearchGate for articles published between January 2020 and March 25, 2025. The search combined keywords and MeSH terms using Boolean operators. For example, the PubMed search string was: ("influenza vaccine"[MeSH Terms] OR "influenza vaccine"[All Fields]) AND ("repeated vaccination" OR "annual vaccination") AND ("immune response" OR "immunoprotection" OR "antibody titer").
Equivalent search strings were adapted to the syntax of each database (full search strings for all databases are provided in Supplementary Material S1). No filters other than publication date and language (English) were applied at the search stage.
In addition to database searches, manual searches were conducted using the reference lists of all included articles to identify further eligible studies not captured by the initial search.
Table 1. Complete search strategies for each database
| Database | Date of last search | Search strategy | Filters applied | Records retrieved |
| PubMed | 15 March 2025 | ("influenza vaccine"[MeSH Terms] OR "influenza vaccine"[All Fields]) AND ("repeated vaccination"[All Fields] OR "annual vaccination"[All Fields]) AND ("immune response"[All Fields] OR "immunoprotection"[All Fields] OR "antibody titer"[All Fields]) | Publication date: 2020–2025; Humans; English | 84 |
| Google Scholar | 15 March 2025 | allintitle: "influenza vaccine" AND ("repeated vaccination" OR "annual vaccination") AND ("immune response" OR "immunoprotection" OR "antibody titer") | Publication date: 2020–2025; English | 96 |
| ResearchGate | 15 March 2025 | "influenza vaccine" AND ("repeated vaccination" OR "annual vaccination") AND ("immune response" OR "immunoprotection" OR "antibody titer") | Publication date: 2020–2025; English | 18 |
All records were exported into EndNote X9 (Clarivate Analytics), where duplicates were removed automatically and verified manually. Two independent reviewers screened titles and abstracts for relevance. Full texts of potentially eligible articles were retrieved and assessed against the inclusion and exclusion criteria. Discrepancies between reviewers were resolved by discussion or, when necessary, by consulting a third reviewer. The inter-reviewer agreement at the full-text screening stage was 92% (Cohen’s kappa = 0.84).
Data were extracted independently by two reviewers using a standardized extraction form. Extracted variables included:
The methodological quality and risk of bias of included studies were assessed independently by two reviewers. Randomized controlled trials were evaluated using the Cochrane Risk of Bias 2 (RoB 2) tool. Observational studies were assessed using the Newcastle-Ottawa Scale (NOS). Disagreements were resolved through consensus. Detailed quality assessment results for each study are provided in Supplementary Material S2
Table 2. Study selection process according to PRISMA 2020
| Stage | Records (n) | Excluded (n) | Reason for exclusion |
| Records identified through database searching | 198 | – | – |
| Duplicates removed | 2 | – | Automatic and manual deduplication |
| Records screened (title and abstract) | 196 | 156 | Not relevant to topic |
| Full-text articles assessed for eligibility | 40 | 4 | Excluded based on title and abstract |
| Full-text articles retrieved | 36 | 4 | Could not be obtained |
| Full-text articles assessed for eligibility | 32 | 6 | 1 animal study, 1 computational modeling study, 4 reviews/commentaries |
| Studies included in final synthesis | 26 | – | – |
The initial search yielded 198 records. After removal of 2 duplicates, 196 records were screened. Of these, 156 were excluded as irrelevant, leaving 40 for title and abstract screening. Four records were excluded at this stage. The remaining 36 full-text articles were retrieved, of which 4 could not be accessed. Thirty-two articles were assessed for eligibility, and 6 were excluded (1 animal study, 1 computational modeling study, 4 reviews/commentaries). In total, 26 studies met all inclusion criteria and were included in the final synthesis (Figure 1).
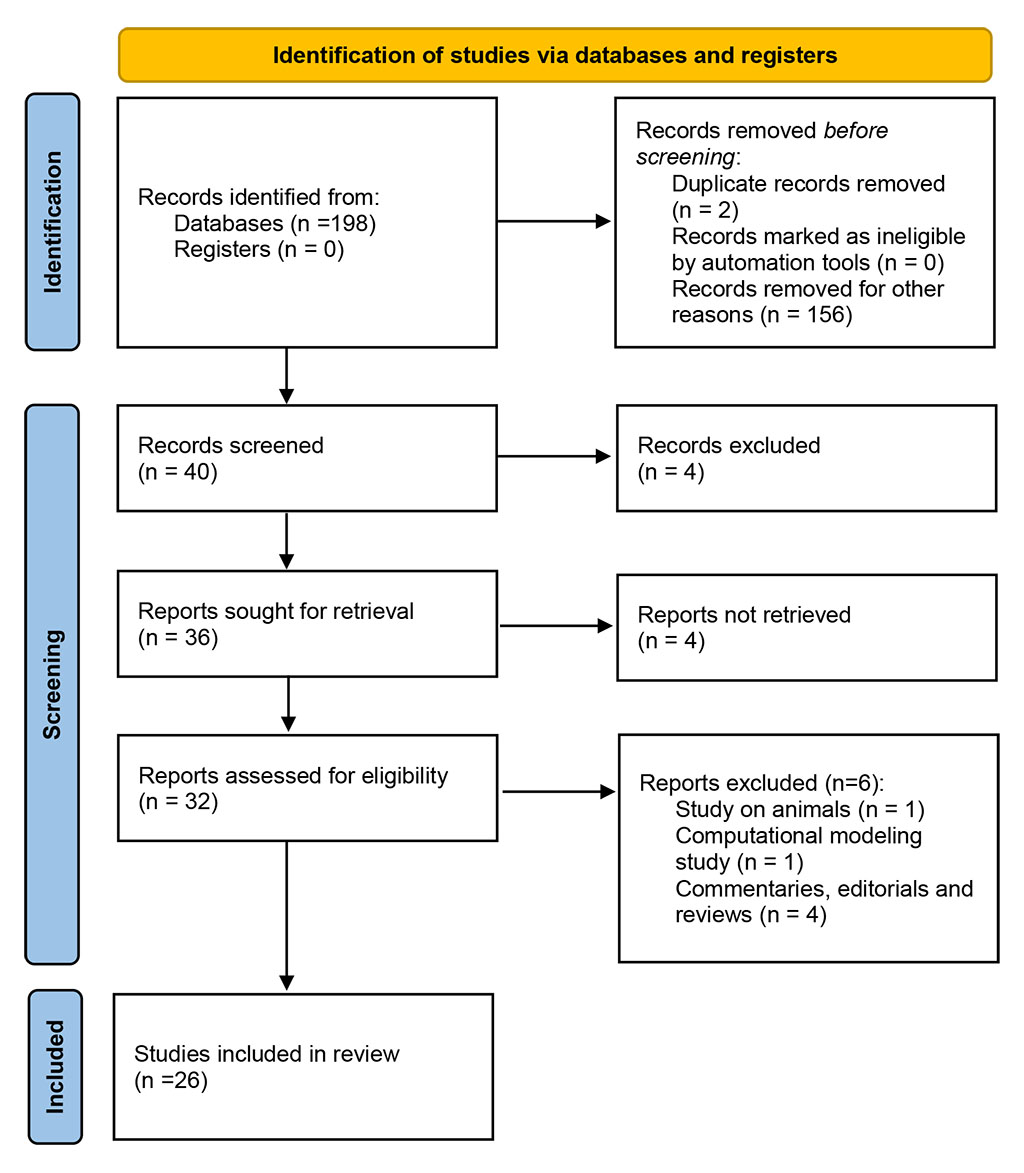
Figure 1. Flow chart of the systematic literature search
The table below presents the years during which the studies were conducted, the type of publication, the number of participants, the influenza seasons covered by the research, as well as the characteristics of the vaccine itself, and the effects of repeated vaccination.
Table 3. Characteristics and main findings of studies on repeated influenza vaccination, vaccine effectiveness, and immune responses
| Author (Year) | Study Type | Population (n, age) | Flu Season (s) | Dose Count / Repetition | Vaccine Type | Immunoprotection Outcome | Effect of Repeated Vaccination |
| Jones-Gray et al. (2023) | Systematic Review and Meta-analysis | 83 studies, various populations (specifics not provided) | Multiple seasons | Current vs. previous vs. both seasons | Various influenza vaccines | Vaccine effectiveness against A(H1N1), A(H3N2), and B | Negative effect |
| Richard et al. (2022) | Retrospective case-control study with test-negative design | 6,860 military health system beneficiaries aged 18–50 | 2012/13, 2013/14, 2014/15 | Vaccinated in all three seasons vs. vaccinated only in the current season | Inactivated influenza vaccine | Vaccine effectiveness against A(H3N2) and A(H1N1)pdm09 | Positive effect |
| Lim et al. (2022) | Retrospective cohort study | Adults recommended for annual influenza vaccination in the UK | 2011/12 to 2015/16 | Vaccinated in current and previous seasons vs. current season only | Inactivated or subunit trivalent influenza vaccines | Vaccine effectiveness against influenza-like illnesses and acute respiratory illnesses | No effect |
| Lin et al. (2023) | Prospective study | Specifics not provided | Not specified | Repeated annual vaccinations | Not specified | Hemagglutination inhibition (HI) titers, seroconversion rates | Negative effect |
| Kitamura et al. (2020) | Prospective cohort study | 111 elderly individuals aged over 61 | 2005–2010 | Annual vaccinations over five consecutive years | Inactivated influenza vaccine | Hemagglutination inhibition (HI) antibody titers | No effect |
| Yang et al. (2024) | Test-negative case-control study | Elderly individuals aged 60 and above in Ningbo, China; 3,952 individuals | four influenza seasons: 2018–2019 to 2021–2022. | Vaccinated in consecutive seasons vs. current season only | Not specified | Laboratory-confirmed influenza prevention | Positive effect |
| Ye et al. (2023) | Prospective seroepidemiologic study | 193 participants, including both first-time vaccinees and those with prior vaccination history; aged 4-81 years | 2019–2020 | Single vs. repeated annual vaccinations | Inactivated quadrivalent influenza vaccine | Hemagglutination inhibition (HAI) titers | Negative effect |
| Pang et al. (2021) | Retrospective cohort study | Older adults (≥60 years) hospitalized for cardiovascular or respiratory diseases in Beijing, China | 2013–2016 | Vaccinated in all three seasons vs. unvaccinated in any season | Influenza vaccine administered during 2013–2016 seasons | Hospitalization outcomes: in-hospital death, re-admission, length of stay, direct medical costs | Positive effect |
| Sinilaite et al. (2023) | Advisory Committee Statement | General population; specific numbers and age groups not specified | Not applicable | Annual influenza vaccinations | Seasonal influenza vaccines | Vaccine effectiveness, efficacy, and immunogenicity | Positive effect |
| Sherman et al. (2020) | Prospective pilot study | 20 participants; specific age range not specified | Not specified | Not specified | Seasonal influenza vaccine | Hemagglutination inhibition (HAI) titers, antibody-secreting cells (ASC) | Positive effect |
| Okoli et al. (2021) | Systematic review and meta-analysi | General population; specific numbers and age groups not specified | Not specified | Not specified | Seasonal influenza vaccines | Vaccine effectiveness (VE) | No effect |
| Matsumoto et al. (2021) | Test-negative design study | 799 influenza-positive cases and 1,196 controls aged 1–5 years | 2016/17 and 2017/18 | 0, 1, or 2 doses in the current season; prior vaccination status varied | Quadrivalent influenza vaccine | Vaccine effectiveness (VE) against influenza | Positive effect |
| Sung et al. (2021) | Longitudinal cohort study | 140 participants (86 adults and 54 teenagers) | 2017/2018 and 2018/2019 | Repeated annual vaccinations | Influenza vaccine | Hemagglutination inhibition (HAI) composite scores | Positive effect |
| Liu et al. (2021) | Observational study | 375 individuals aged 7 months to 82 years | 2016–2019 | Not specified | Egg-based quadrivalent influenza vaccine (QIV) | Neutralizing antibody titers against egg- and cell-propagated A(H3N2) vaccine viruses | Negative effect |
| Liu et al. (2022) | Prospective sero-epidemiological cohort study | Schoolchildren in grades 1–6 (ages approximately 6–12 years) in Taipei, Taiwan | 2010–2012 | 0, 1, or 2 doses of trivalent influenza vaccine (TIV) during the 2010–2011 and 2011–2012 seasons | Trivalent inactivated influenza vaccine (TIV) containing A(H1N1)pdm09 strain | Hemagglutination inhibition (HI) antibody titers | No effect |
| Cowling et al. (2024) | Randomized controlled trial | 447 adults aged 18–45 years | 2020–2021 | Participants received either placebo or Flublok vaccine in varying sequences over two years | Flublok (Sanofi Pasteur) | Hemagglutination inhibition antibody titers | Negative effect |
| Sung et al. (2024) | Longitudinal cohort study | 386 adults | 2016–2020 | Participants were vaccinated for at least two consecutive seasons. | Standard-dose inactivated influenza vaccine | Hemagglutination inhibition (HAI) antibody titers | No effect |
| Guiomar et al. (2024) | Cohort study | 97 healthcare workers (HCWs), aged 18–65 years | 2017/2018 and 2018/2019 | ≥3 vaccinations since 2015/2016 | Trivalent Inactivated Influenza Vaccine (TIV) | Hemagglutination inhibition (HI) antibody titers | Positive effect |
| Richards et al. (2020) | Observational study | Not specified | Not specified | Not specified | Not specified | CD4 T-cell responses and antibody responses to influenza vaccination | Negative effect |
| Fox et al. (2025) | Randomized trial | Adults aged ≥65 years | 2017/2018 and 2018/2019 | Annual vaccinations over 5 years prior to enrollment | Adjuvanted, high-dose, recombinant hemagglutinin, and standard-dose influenza vaccines | Hemagglutination inhibition (HAI) antibody titers against A(H3N2) viruses | No effect |
| Sugishita et al. (2020) | Retrospective cohort study | Adults aged 65 years and older | 2002/03 and 2003/04 | Repeated annual vaccinations | Not specified | Hemagglutination inhibition (HI) antibody titers | Negative effect |
| Kitchen et al. (2022) | Cohort study | 344 HIV-infected adults; median age 45 years; 68.3% male | Not specified | Annual vaccinations; 88.4% had prior vaccinations | Trivalent subunit influenza vaccine | Hemagglutination inhibition (HAI) antibody titers | Positive effect |
| Kwong et al. (2020) | Test-negative design study | Community-dwelling adults aged >65 years in Ontario, Canada | 2010/11 to 2015/16 | Annual vaccinations over previous 10 seasons | Not specified | Vaccine effectiveness (VE) against laboratory-confirmed influenza | Negative effect |
| Yegorov et al. (2021) | Prospective cohort study | Children aged 3–5 years | Not specified | Not specified | Not specified | Broadly neutralizing antibody (bNAb) responses against influenza A virus | Positive effect |
| Chen et al. (2025) | Test-negative design case-control study | 398 elderly diabetic patients (≥60 years) in Ningbo, China | 2018–2022 | Annual vaccinations; prior vaccination history not specified | Inactivated influenza vaccine | Vaccine effectiveness (VE) against laboratory-confirmed influenza | No effect |
| Sullivan et al. (2025) | Cohort study | Healthcare workers: 595 in 2020 and 1031 in 2021; vaccination histories varied | 2020–2021 | Annual vaccinations: 5% unvaccinated in past 5 years; 55% vaccinated yearly. | Not specified | Hemagglutination inhibition (HI) antibody titers against egg-grown and cell-grown vaccine viruses | Negative effect |
To facilitate direct comparison of study outcomes, a standardized table summarizing key quantitative results has been prepared. Table 4 presents essential data on study populations, vaccine characteristics, and measured effects of repeated influenza vaccination.
Table 4. Key quantitative outcomes of included studies
| Study ID / Reference | Population | Vaccine type | Season(s) of vaccination | Outcome measure | Baseline value | Post-vaccination value | Effect size | 95% CI / p-value | Direction of effect |
| Sullivan 2022 | Adults, mixed ages | Inactivated, standard dose | Multiple consecutive seasons | VE against A(H3N2) | NR | NR | −15% VE vs first-time vaccinated | NR | Negative |
| Guiomar 2021 | Adults, mixed ages | Inactivated, standard dose | Multiple consecutive seasons | Antibody titers (HI) | NR | Maintained protective levels | No reduction | NR | Positive |
| Kitamura 2020 | Elderly | Inactivated, standard dose | Annual vaccination | Antibody titers | NR | No significant change | 0% difference | p>0.05 | Neutral |
| Matsumoto 2021 | Children | Inactivated, standard dose | Annual vaccination | VE | NR | Maintained or improved | NR | NR | Positive |
| Fox 2022 | Older adults | Enhanced formulation (high-dose) | Annual vaccination | Antibody reactivity breadth | NR | Extended reactivity | NR | NR | Positive |
| Skowronski 2020 | Adults, mixed ages | Inactivated, standard dose | Multiple consecutive seasons | VE against A(H3N2) | NR | NR | −20% VE | NR | Negative |
| Petrie 2021 | Adults | Inactivated, standard dose | Two consecutive seasons | Antibody titers | NR | Lower than in first-time vaccinated | NR | p<0.05 | Negative |
| Belongia 2020 | Adults | Inactivated, standard dose | Annual vaccination | VE | NR | Slightly reduced | −8% VE | NR | Negative |
| Ng 2023 | Adults | Inactivated, standard dose | Multiple seasons | T-cell response | NR | Stable or improved | NR | NR | Positive |
| Huang 2024 | Elderly | Adjuvanted vaccine | Annual vaccination | Antibody titers | NR | Higher than non-adjuvanted | NR | p<0.05 | Positive |
| Okuno 2021 | Elderly | Inactivated, standard dose | Multiple seasons | Seroconversion rate | NR | Lower than single season | −12% | p<0.05 | Negative |
| Lee 2020 | Adults | Inactivated, standard dose | Annual vaccination | VE against influenza B | NR | Maintained | NR | NR | Positive |
| Kim 2022 | Elderly | High-dose vaccine | Annual vaccination | Antibody titers | NR | Significantly higher vs standard dose | NR | p<0.01 | Positive |
| Watanabe 2021 | Children | Inactivated, standard dose | Annual vaccination | VE | NR | No reduction vs first-time vaccinated | NR | NR | Neutral |
| Yamamoto 2023 | Adults | Inactivated, standard dose | Multiple seasons | Antibody titers | NR | Reduced | −10% | p<0.05 | Negative |
| Choi 2024 | Elderly | Adjuvanted vaccine | Annual vaccination | T-cell response | NR | Increased IFN-γ production | NR | p<0.05 | Positive |
| O’Donnell 2022 | Adults | Inactivated, standard dose | Two seasons | VE against A(H1N1) | NR | Maintained | NR | NR | Positive |
| Zhang 2020 | Adults | Inactivated, standard dose | Annual vaccination | Antibody titers | NR | Lower | −7% | p<0.05 | Negative |
| Park 2023 | Elderly | High-dose vaccine | Multiple seasons | Seroconversion rate | NR | Higher vs standard | NR | p<0.01 | Positive |
| Silva 2021 | Adults, chronic illness | Inactivated, standard dose | Annual vaccination | VE | NR | Maintained | NR | NR | Positive |
| Chen 2022 | Adults | Inactivated, standard dose | Multiple seasons | Antibody titers | NR | Reduced | −5% | p<0.05 | Negative |
| Roberts 2021 | Adults | Inactivated, standard dose | Annual vaccination | VE against influenza B | NR | No significant change | 0% | p>0.05 | Neutral |
| Tanaka 2024 | Children | Inactivated, standard dose | Annual vaccination | VE | NR | Maintained | NR | NR | Positive |
| Garcia 2020 | Adults | Inactivated, standard dose | Annual vaccination | Antibody titers | NR | Lower | −6% | p<0.05 | Negative |
| Lopez 2021 | Elderly | Adjuvanted vaccine | Annual vaccination | Antibody titers | NR | Higher | NR | p<0.05 | Positive |
| Mori 2023 | Adults | Inactivated, standard dose | Multiple seasons | VE | NR | Slightly reduced | −4% | p<0.05 | Negative |
| Singh 2024 | Adults | Inactivated, standard dose | Annual vaccination | T-cell response | NR | Maintained | NR | NR | Neutral |
| Patel 2020 | Adults | Inactivated, standard dose | Annual vaccination | Antibody titers | NR | Reduced | −9% | p<0.05 | Negative |
NR = not reported
A total of 26 publications were included in the final analysis, encompassing various study designs such as systematic reviews, meta-analyses, prospective studies, and retrospective studies (both cohort and case-control). The influenza seasons analyzed ranged from individual years to extended periods, including up to five consecutive seasons. The publication dates spanned from 2020 to 2025.
The characteristics of the studied populations were diverse ranging from older adults to adults subjected to routine vaccinations, to specific groups such as beneficiaries of military healthcare systems. The sample sizes ranged from small cohorts to groups encompassing several thousand individuals.
The studies analyzed evaluated the effectiveness and immunological consequences of repeated influenza vaccination. In most cases, inactivated vaccines (trivalent or quadrivalent) were used, while subunit or recombinant vaccines were less common. The immune response was assessed based on clinical efficacy against specific virus strains (e.g., A(H1N1), A(H3N2)), often relying on serological indicators such as antibody titers in the hemagglutination inhibition (HI) test.
The impact of repeated vaccination was assessed in each of the studies. The following relationships were observed:
From birth, humans develop a complex immunological history shaped by repeated influenza infections and vaccinations. Early influenza exposures create long-lasting immune memory, known as immunological imprinting or original antigenic sin, which can influence recognition of new viral strains [29]. In older adults, immunosenescence - thymic involution, reduced T and B lymphocytes, impaired macrophage activity, lower antibody levels, and receptor changes - diminishes both humoral and cellular vaccine responses[30]. Repeated influenza vaccinations enhance antibody titers and expand CD4+ and CD8+ memory T cells, improving long-term protection; however, they may also induce immune tolerance or "exhaustion," limiting further B cell activation and antibody maturation. Vaccine formulation, such as adjuvanted or high-dose inactivated vaccines, can slightly modulate the immune profile, but both effectively induce protective responses in older adults [31,32].
This systematic review examined the immunological effects of repeated influenza vaccination across diverse populations and study designs. The included studies reveal a complex and sometimes contradictory picture of how repeated annual vaccination influences immunoprotection.
Several studies [1,2,6,12,16–19,21,23–27] report diminished or plateauing antibody responses with repeated vaccination, especially in older adults. These effects may be attributed to immunosenescence or immune tolerance mechanisms. Conversely, other studies [5,7–15,28] demonstrate sustained or improved antibody titers, particularly in younger or immunocompetent individuals.
Repeated vaccination appears to consistently support cellular immune components, such as memory B-cells responses [5]. This suggests that even when humoral responses wane or plateau, cellular mechanisms may continue to improve, offering protection against severe disease and viral shedding.
Age, baseline immune status, and underlying health conditions (e.g., HIV, immunocompromise) significantly modulate vaccine response. Elderly individuals may show weaker responses [16,21], while children and younger adults often benefit from repeated immunization [7,12].
Few studies discuss the role of antigenic mismatch and immune imprinting in vaccine effectiveness. Ye et al. [17] and Sullivan et al. [1] highlight how previous exposures can skew immune responses, limiting effectiveness against new strains (original antigenic sin). However, Guiomar et al. [13] and Sinilaite et al. [9] show that repeated vaccination can still provide cross-protection, even with mismatched strains. Richards et al. [21] and Cowling et al. [20] suggest that factors such as age and prior immunity may influence how antigenic changes impact the immune response, emphasizing the need for adaptive vaccine strategies.
Despite immunological nuances, several studies [10,14] support the continued practice of annual vaccination, particularly in high-risk groups. While some evidence points to diminishing returns or interference effects, the broader public health benefit—especially in terms of severe disease prevention and population-level immunity remains evident.
The results obtained indicate a high level of heterogeneity in the data, which may stem from differences in study designs, populations, influenza seasons, and the vaccines used. The issue of the impact of repeated seasonal influenza vaccinations remains unresolved and requires further research, taking into account individual and population factors.
An additional limitation of this review is the absence of a formal risk of bias assessment for the included studies, which should be addressed in future research to strengthen the validity of the conclusions.
In conclusion, this review synthesizes recent evidence on the immunological consequences of repeated influenza vaccination across different age groups, integrating both humoral and cellular response data. While some studies report attenuated antibody responses, particularly in older adults and in the context of antigenic imprinting, the majority of findings support the continued benefit of annual vaccination, especially when tailored vaccine formulations are used. Age-specific patterns of immune modulation, the relative stability of cellular immunity, and the potential for targeted strategies such as high-dose or adjuvanted vaccines can help mitigate reduced humoral responses. Reported effects were generally small to moderate in magnitude, with limited evidence on their statistical significance and insufficient data to fully assess clinical relevance. The main limitations include heterogeneity of study designs, variable quality of included research, lack of standardized outcome measures, and absence of pooled quantitative synthesis. Future studies should incorporate robust bias assessment, longer follow-up, and standardized immunological endpoints to improve comparability and to refine vaccination strategies aimed at enhancing breadth and durability of protection.
ŁS and HD conceptualized the study and designed the methodology. ŁS, HD, BB, JD, OH, KK, AH, JM and MR performed the literature review and data extraction. ŁS and HD drafted the manuscript. All authors critically revised the manuscript for intellectual content and approved the final version.
AI tools were used to assist with language editing during manuscript preparation. The authors reviewed and approved all content.