- Home
- About the Journal
- Peer Review
- Editorial Board
- For Authors
- Reviewer Recognition
- Archive
- Contact
- Impressum
- EWG e.V.
![]() alekseymichailovich22976@gmail.com
alekseymichailovich22976@gmail.com
Hypothyroidism causes a number of histological changes in almost all tissues and cells of the body of a mammal, which is accompanied by numerous clinical and functional disorders. This is due to the multifaceted and multifunctional role of thyroid hormones (thyroxine and triiodothyronine) in the body of mammals. Striated cardiac muscle tissue and its main structural unit (contractile cardiomyocyte) is one of the most frequently involved tissues in the histopathological process in hypothyroidism. In addition, hypothyroidism can be considered as one of the main risk factors for the development of cardiovascular diseases (heart failure, coronary heart disease, cardiac arrhythmias), so the study of histopathological changes in cardiac muscle tissue is of great theoretical and practical importance. To date, there are no studies evaluating histopathological ultrastructural changes in contractile cardiomyocytes in certain forms of hypothyroidism, in particular in overt acquired primary hypothyroidism. The purpose of this study is to investigate the histopathological changes in the nucleus and contractile apparatus of cardiomyocytes in the conditions of experimental modeling of overt acquired primary hypothyroidism. Materials and Methods: male Chinchilla rabbits aged 12-14 months and weighing 3.1-3.5 kg (n=10), were divided into 2 equal groups. 1) The experimental group (n=5), where overt primary hypothyroidism was induced by oral administration of the antithyroid drug thiamazole (10 mg/kg for 4 weeks). 2) The animals of the control group (n=5) received placebo and were in a state of normothyroidism. Transmission electron microscopy (TEM) was used to study ultrastructural changes in contractile cardiomyocytes. Results and Conclusion: Based on the results of electron microscopic study, we detected numerous ultrastructural changes in the nucleus and contractile apparatus of cardiomyocytes: the nucleus (wrinkling, uneven contours of the nucleus and emergence of karyolemma invaginations, chromatin condensation); in the contractile apparatus (thinning of myofibrils, emergence of areas of myofibrils overcontraction with contractures). These ultrastructural changes in cardiomyocytes underlie the disruption of myocardial contractility, as well as an increase in dystrophic and apoptotic changes with the gradual development of heart failure, which is a frequent companion of untreated overt acquired primary hypothyroidism.
Keywords: primary hypothyroidism, experimental modeling, cardiomyocytes, nucleus, contractile apparatus
Hypothyroidism is manifested by pronounced morphological and clinical changes affecting almost all organs and tissues, due to the presence of multiple targets for thyroid hormones and their multifaceted and multifunctional role in the body of mammals [1–3]. In a significant number of patients, hypothyroidism is mild, manifesting itself only with nonspecific subclinical signs that may not bother patients for a long time, but can induce a number of irreversible changes and complications, up to death [1]. Thus, according to clinical studies, hypothyroidism causes the development of heart failure and increases the risk of adverse events [4, 5]. And according to the experimental study by Japanese scientists (Y Ono et al), untreated hypothyroidism in rabbits during 3 months of observation leads to death in 6 out of 7 individuals from developing heart failure [6]. Due to the wide variety of clinical manifestations and the general lack of specificity of symptoms, the diagnosis of hypothyroidism is based on clinical laboratory criteria. So, overt or clinically apparent primary hypothyroidism is diagnosed if the concentration of thyroid-stimulating hormone of the adenohypophysis (TSH) is above the reference range, and the concentration of free thyroxine is below the reference range. Mild or subclinical hypothyroidism, which is usually considered as a sign of early thyroid insufficiency, and is diagnosed if the concentration of TSH is above the reference range, and the concentration of thyroxine and triiodothyronine is within the normal range [1, 3]. Thus, the most reliable and optimal method for diagnosing hypothyroidism is clinical and laboratory diagnostics based on determining the concentration of thyroid hormones and adenohypophysis in the blood serum of patients. The classification of hypothyroidism is based on three main principles: the time of development, the cause of occurrence and the severity [1-3] (Table 1).
Table 1. Hypothyroidism classification criteria
| 1. Time of development | 2. Cause of occurrence | 3. Severity level |
| Congenital |
Primary (thyroid) | Mild or subclinical |
| Secondary (pituitarium) | Overt (manifest) or clinically apparent | |
| Acquired | Tertiary (hypothalamic) | Complicated or severe |
| Peripheral (tissue) |
The most common forms of hypothyroidism in actual clinical practice are primary acquired hypothyroidism of clinical and subclinical degrees of severity [1-3]. Recently, morphological studies of histopathological changes in organs and tissues of mammals under induced hypothyroidism have become highly relevant in fundamental medicine [6–9]. According to a number of morphological studies, hypothyroidism causes significant structural changes in the organs and tissues of the cardiovascular system [7-14], the skin and its derivatives [15, 16], the musculoskeletal system [17, 18], and the reproductive system [19, 20]. To date, a number of morphological studies of the myocardium at the tissue level of organization have been carried out. However, there are no studies evaluating the histopathological changes in the myocardium, in particular its structural unit (contractile cardiomyocytes) at the subcellular (ultrastructural) level of organization with certain degrees of forms of hypothyroidism, in particular, taking into account severity of hypofunction of the thyroid gland. Contractile cardiomyocytes are the main and most numerous cellular components of striated cardiac muscle tissue and are most often involved in pathogenesis of cardiac and extracardiac pathologies, which are accompanied by various disorders of myocardial contractile function. The study of ultrastructural changes in contractile cardiomyocytes and understanding of the mechanisms underlying their development is necessary for the search and development of solutions on neutralizing them while optimizing the management of patients with hypothyroidism. At the same time, taking into account a quite significant number of forms of hypothyroidism and the difference in the mechanisms of their etiopathogenesis, both clinical and morphological studies should be carried out for each form in order to search for specific signs that could be used to optimize patient care.
The purpose of this study is to investigate and describe the histopathological changes in the nucleus and contractile apparatus of cardiomyocytes under experimentally induced overt acquired primary hypothyroidism.
The object of the study was male Chinchilla rabbits aged 12-14 months and weighing 3.1-3.5 kg (n=10). Animals were divided into 2 equal groups: 1) experimental (n=5), in which experimental modeling of primary hypothyroidism of manifest severity was carried out by oral administration of the antithyroid drug thiamazole, which inhibits the formation of thyroid hormones, through a tablet dispenser at a dose of 10 mg/kg daily during 4 weeks (Figure 1); 2) control (n=5), who received placebo and were in a state of normothyroidism. The animals were kept in the vivarium of the Institute of Experimental Medicine and Biotechnology of the Samara State Medical University, they were cared for in accordance with the rules and regulations for handling laboratory animals, in accordance with the "International recommendations for conducting biomedical research using animals" (1985), with the rules of laboratory practice in the Russian Federation (Order of the Ministry of Health of the Russian Federation dated 19.06.2003 No. 267) and the Law on Protection of Animals from Cruelty, Chapter V, art. 104679-GD dated 01.12.1999. The permission of the bioethics committee at the Samara State Medical University was obtained to conduct the study (protocol No. 204 dated 11.12.2019).
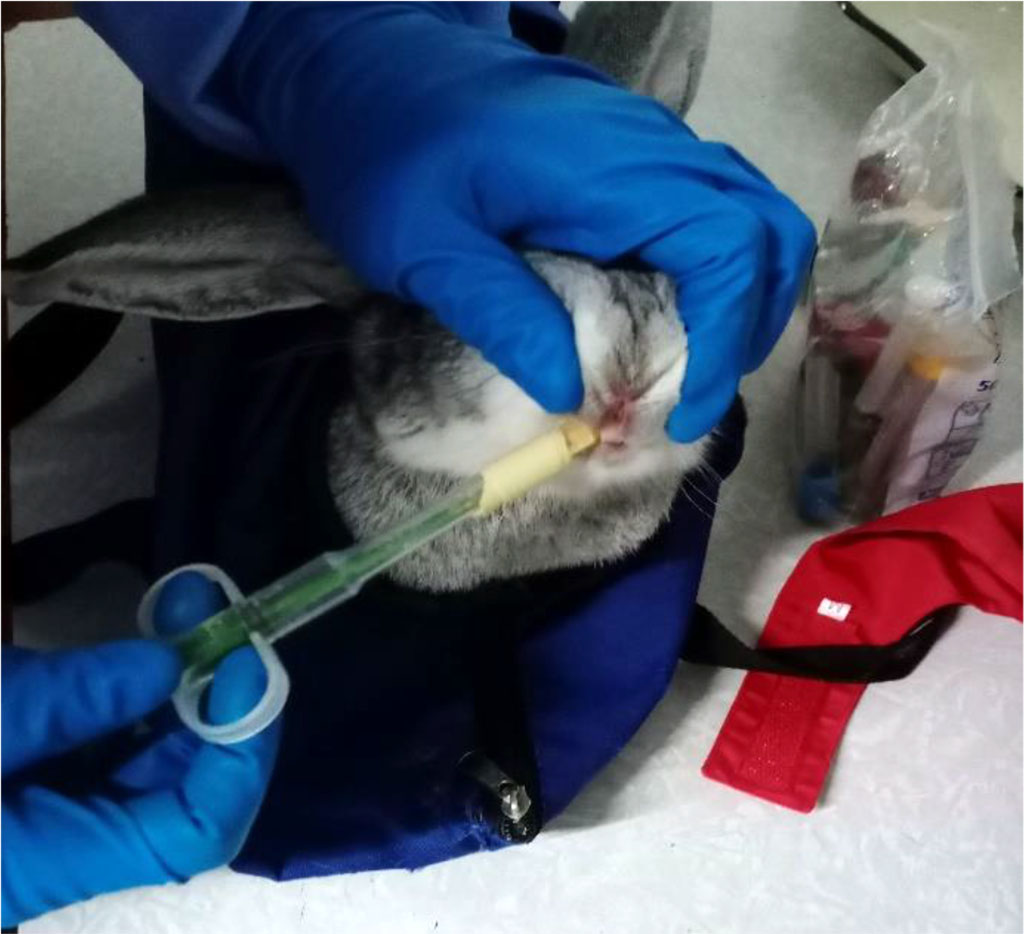
Figure 1. Primary hypothyroidism medicated modeling
To confirm the development of hypothyroidism in all animals, at the end of the modeling period, blood was taken from the ear vein, and after centrifugation, blood serum was obtained. An enzyme-linked immunosorbent assay was then used for the rabbit serum samples to determine the concentration of thyroid hormones (total triiodothyronine (Fine Test, cat. no. EU0403)), total thyroxine (Cusabio, cat. no. CSB-E06936Rb)) and adenohypophysis TSH (Cusabio, cat. no. CSB-E06916Rb). In the course of our experiments, the animals did not receive additional drugs to compensate for the state of thyroid hypofunction. On the 29th day, the animals were taken out of the experiment in order to take myocardial samples for electron microscopic examination. Transmission electron microscopy (TEM) was used to study and describe the ultrastructural changes in contractile cardiomyocytes. Myocardial biomaterial from the anterior wall of the left ventricle was then fixed in the solution of glutaraldehyde in phosphate buffer, embedded in araldite, ultrathin sections were contrasted with the 2.5% solution of uranyl acetate, and then the sections were examined using a Hitachi TEM System electron microscope at the Kazan (Privolzhsky) Federal University (Kazan, Russia).
When laboratory rabbits are kept on an antithyroid drug (thiamazole) at the dose of 10 mg/kg for 4 weeks, overt primary hypothyroidism develops, which, in accordance with current international and Russian clinical guidelines for hypothyroidism [1, 3], manifests itself as a decrease in the concentration of triiodothyronine and thyroxine and an increase in the concentration of TSH compared with the control group (Table 2). Thus, the development of overt hypothyroidism in laboratory rabbits was confirmed by the results of the clinical and laboratory study.
Table 2. Concentration of hormones in rabbits of the control and experimental groups
| Lab parameter | Experimental group, n=5 | Control group, n=5 | Test of validity, p |
| TSH, μIU/ml | 6,384±0,575 | 0,572±0,067 | <0,001 |
| Total triiodothyronine, ng/ml | 0,558±0,042 | 1,610±0,299 | <0,001 |
| Total thyroxine, ng/ml | 3,504±0,346 | 9,407±1,203 | <0,001 |
A change in the structure of the nucleus, characterized by an uneven contour of the nuclear membrane, in manifest primary hypothyroidism is a very characteristic phenomenon and was found in electron diffraction patterns of all animals of the experimental group. The contours of the nuclear membrane became tortuous due to the appearance of invaginations, and the shape of the nucleus itself became more wrinkled. Karyolemma invaginations ranged from shallow to very pronounced (Figure 2), while in the control group, the contours of the nucleus were smooth or had a relatively small number of shallow karyolemma invaginations. In a small number of cardiomyocytes, the invaginations of the nuclear membrane were so deep that it gave the impression of a gradual physical separation of the nucleus into separate fragments. Very characteristic changes in hypothyroidism were changes in the structure of the nucleus, expressed in the redistribution of chromatin and its condensation. In the nucleus of cardiomyocytes of intact animals, darker areas (heterochromatin), localized on the periphery in the form of the thinnest rim, and a lighter area (euchromatin), occupying a central position, are noticeable. In the euchromatin area, occasional finely punctate zones of heterochromatin were found. Whereas in the nuclei of cardiomyocytes of animals in the state of hypothyroidism, the heterochromatin rim was more pronounced and in the center of euchromatin an increase in the number and size of heterochromatin areas was noted. Around some nuclei of contractile cardiomyocytes a slight perinuclear edema was found, which is not typical for the nuclei of contractile cardiomyocytes of the intact group of rabbits. According to recent studies, a change in the contours of the nucleus, wrinkling of the nucleus and condensation of chromatin is one of the early signs of cell apoptosis. Thus, our data on structural changes in the nuclei of contractile cardiomyocytes in hypothyroidism are consistent with other researchers, who, using molecular genetic research methods, found activation of proapoptotic and suppression of antiapoptotic signaling pathways and enzymes in a number of cells (cardiomyocytes, neurocytes, macrophages, smooth myocytes, hepatocytes, etc.) in case of hypothyroidism [21-26].
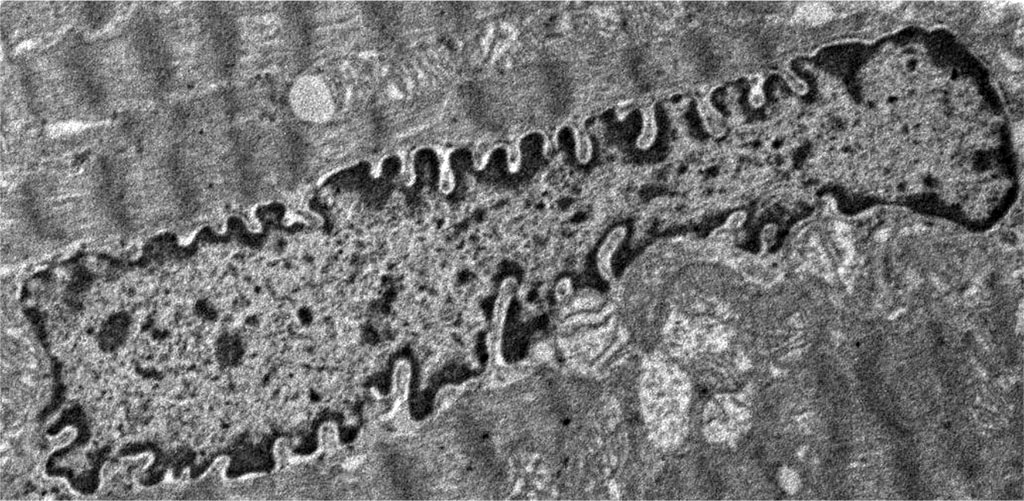
Figure 2. Histopathological changes in the nucleus of contractile cardiomyocytes: overt primary hypothyroidism on the 29th day from the start of the experiment. Magnification 2400. TEM.
Very noticeable ultrastructural changes were found in the contractile apparatus of contractile cardiomyocytes. In animals of the experimental group, thinning of myofibrils was noted, compared with animals in the control group. The characteristic feature was the presence of areas of overcontraction of myofibrils, characterized by a decrease in the size of sarcomeres (a decrease in the distance between telophragms), a decrease in the size of light discs and the H-zone around the mesophragm (Figure 2 and Figure 3).
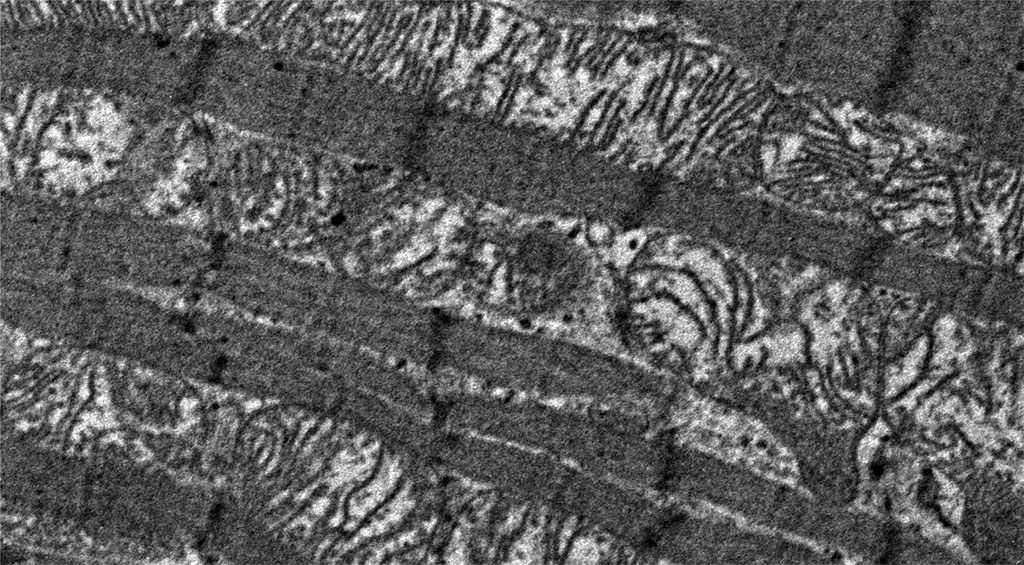
Figure 3. Histopathological changes in myofibrils of contractile cardiomyocytes: overt primary hypothyroidism on day 29 from the start of the experiment. Magnification 4200. TEM.
In some areas of contractile cardiomyocytes, the overcontractions of myofibrils were so pronounced that they led to the formation of areas of pronounced overcontraction or contractures (Figure 4), while these changes were absent in the control group. The visually recorded thinning of myofibrils in hypothyroidism may be due to a decrease in the formation of myosin protein in contractile cardiomyocytes, as reported in the molecular genetic study by N. Yousefzadeh et al, where the researchers reported a decrease in the expression of the myosin protein isoform characteristic of adults (heavy chain isoform alpha-myosin), in the myocardium of rats under conditions of hypothyroidism. A decrease in the expression of the adult myosin isoform was accompanied by a compensatory increase in the expression of the fetal myosin isoform (beta-myosin heavy chain isoform) and weakening of the contractile function of the heart, in particular, a decrease in the frequency and strength of heart contractions [27, 28]. Similar changes in the qualitative and quantitative composition of myosin protein isoforms and impaired contractile function were also noted in striated skeletal muscle tissue [29]. Hypothetically, it can be assumed that the change in the expression of the myosin protein is associated with the ultrastructural anomalies of the myofibrillar apparatus that we discovered. Contracture disorders of myofibrils, at the time of their formation, can be clinically manifested by cardiac arrhythmias, and subsequently lead to the loss of the ability to contract this area of the cardiomyocyte, which can lead to both a decrease in heart rate and a decrease in cardiac output. At the same time, hypothyroidism is indeed clinically manifested by similar myocardial symptoms [27]. Thus, the histopathological changes we found in the structure of the contractile apparatus are characteristic clinical signs of this endocrine nosology [30, 31].

Figure 4. Histopathological changes in the myofibrils of contractile cardiomyocytes. Areas of overcontraction of myofibrils of contractile cardiomyocytes with the presence of contractures. Magnification 6600. TEM.
Thus, our findings has shown, that the medicated acquired primary overt hypothyroidism causes a number of histopathological changes in contractile cardiomyocytes: in the nucleus (irregularities of the contours of the nucleus with the appearance of invaginations of the nuclear membrane, wrinkling, condensation of chromatin), in the contractile apparatus (thinning of myofibrils, the appearance of areas of overcontraction of myofibrils with contractures). These ultrastructural changes in contractile cardiomyocytes underlie the disruption of myocardial contractility, as well as the increase in dystrophic and apoptotic changes with the gradual development of heart failure, which is a frequent companion of untreated overt acquired primary hypothyroidism.