- Home
- About the Journal
- Peer Review
- Editorial Board
- For Authors
- Reviewer Recognition
- Archive
- Contact
- Impressum
- EWG e.V.
Cite as: Archiv EuroMedica. 2024. 14; 2: e1. DOI 10.35630/2024/14/2.208
The purpose of the study was to show the histopathological features of melanoma of the human choroid, as well as the structures surrounding the tumor, and their significance in predicting the outcome of the tumor for choosing a treatment method. The desire for organ-preserving non-invasive methods of tumor destruction leaves out of attention such risk factors as metastasis, localization and degree of damage to the structures surrounding uveal melanomas. To substantiate the unsafety of local treatment of melanomas, the morphological features of the cellular phenotypes of the tumor and the features of changes in the structures surrounding the tumor on sections of a human eye enucleated according to clinical indications were established. It has been established that the tumor contains not only the well-known spindle-shaped and epithelioid cells. The data obtained complement the information on the cellular phenotypes of ocular melanomas and dictate a revision of the generally accepted classification of these tumors, including fusiform, epithelioid and mixed types of malignant tissues in the structure of the human choroid, adopted in the seventh edition of the Cancer Staging Manual of the American Joint Committee on Cancer (Cancer Staging Manual) of the American Joint Committee on Cancer). Pathological changes were established in the tissues adjacent to the tumor, in the retina and vitreous body, which are responsible for the preservation of visual functions.
Keywords: melanoma, human choroid, histopathology, cellular phenotypes of melanoma, features of tumor blood vessels, metastasis
Treatment of ciliary body tumors due to the difficulties of an accurate clinical diagnosis remains a challenge not only because of the anatomical and topographical features of the localization of melanoma, but also in predicting the outcome of the tumor in most cases [1, 2]. The first biometric classification proposed by J. Shields in 1983 was the basis for the strategy of local tumor destruction depending on the size, divided into three groups - initial, medium and large [3].
Despite the use and expansion of indications for brachytherapy (BT) since the 1970s, and the possibility of local tumor control using imaging studies, the expansion of indications for BT is currently the subject of heated debate. Laver N.V., McLaughlin M.E., Duker J.S. (2010) believe that it is clinically and pathohistologically important to determine which morphological features give the worst prognosis in order to choose the most effective treatment strategy [4]. Despite the use of fine-needle aspiration biopsy (FNA) in combination with cytogenetics and the new classification of uveal malignant melanomas, molecular genetic studies of chromosome 3 status, features of location, size, histopathology, and cytogenetic abnormalities remain important risk factors for melanoma progression.
Monitoring and local control of the tumor, however, is accompanied by metastasis in 50% of cases within 15 years after diagnosis. Simpson E.R. (2004) consider the most important problem in late diagnosis and ineffective treatment of melanomas to be the lack of comprehensive data on the mechanisms of malignancy in ocular melanomas and tumor metastasis [5]. Considering the need to treat melanoma before studying pathomorphological changes, there is an urgent need to have a complete picture of histological changes in eye structures in the context of the development of choroidal melanoma for pathogenetically based measures and targeted effects on damaged structures.
The purpose of the study was to elucidate changes not only in the tumor growth zone, but also in the reactions of the eye structures as a whole, which determined the choice of direction for our research.
Human eye material obtained from five patients examined for decreased vision, eye pain and glaucoma, and the presence of a precipitate of pigment cells in the drainage zone was studied.
The treatment method was complete enucleation of the eye, as a surgical method within the gold standard for clinical indications.
The prepared sections from material embedded in paraffin blocks were stained using hematoxylin and eosin staining according to classical protocols and studied using an OLYMPUS microscope equipped with a PD x25 digital camera and an original proprietary program for morphological processing and production of illustrations. The morphological features of tumors in 5 patients after enucleation of the eye according to clinical indications with the presence of a ciliary body tumor were analyzed.
The macroscopic picture of the ciliary body tumor spread mainly on the lateral surface of the eye and reached the posterolateral surface of the choroid. The largest size of the tumor corresponded to the anterior lateral segment of the eye, which was the reason for the delayed diagnosis of melanoma in the setting of gradually increasing clinical manifestations (Figure 1).
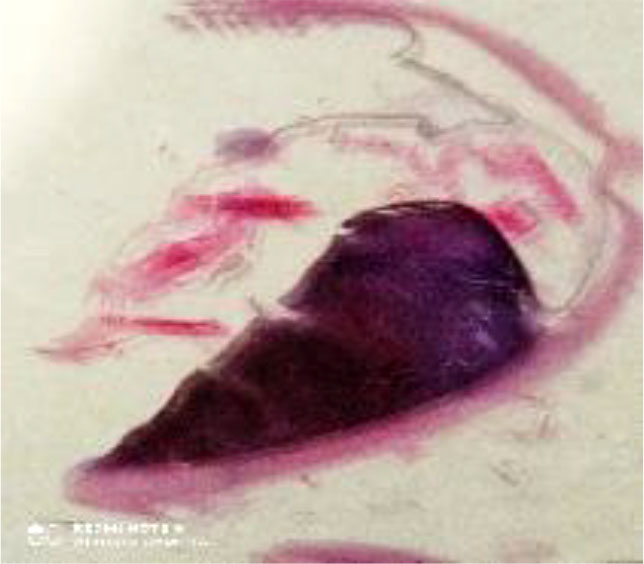
Figure 1. Macroscopic picture of human eye melanoma with a trapezoidal shape. Eye section after enucleation stained with hematoxylin and eosin. The dimensions of the eyeball correspond to the actual dimensions after fixation in neutral formalin.
The wide base of the tumor is adjacent to the outer wall of the eye, without invasion of the sclera, with flexion of Bruch's membrane towards the vitreous. The topography of the tumor corresponds to a lateral location in the ciliary body, in the area where the blind retina is located, with spread to the choroid proper. Such localization is the reason for late diagnosis due to a delay in the decline in visual functions and late presentation of patients.
Currently, there are recognized classifications of choroidal melanomas according to the cell types of spindle-shaped, epithelioid and mixed. Diebold Y., Blanco G., Saornil M.A., et al (1997) presented three cell lines of choroidal melanomas, showing human uveal melanoma cell lines OCM-1, SP 6.5 and MKT-BR, as well as the ocular melanocyte cell line UW-1 with preservation of some structural and ultrastructural characteristics of melanocytic cells. All four MoAbs, PAL-M2, NK1/C3, IND-1 and MAAMA against skin melanoma-associated antigens, stained positively in all melanoma cell lines as well as a melanocytic cell line [6].
In our study, all cell lines exhibited epithelioid/spindle morphology with occasional multinucleated cells and nuclear pleomorphism. However, epithelioid cells according to their shape can be divided into subtypes corresponding to parenchymal epithelial cells and tall prismatic ones. In the structure of ciliary body melanoma, epithelioid cells with hypochromic nuclei, eccentric nucleoli, oxyphilic cytoplasm with transparent vesicles located in the apical part of the cells were identified (Figure 2). The tumor tissue is permeated with cells with hyperchromatic nuclei and chromophobic cytoplasm (Figure 2, D).
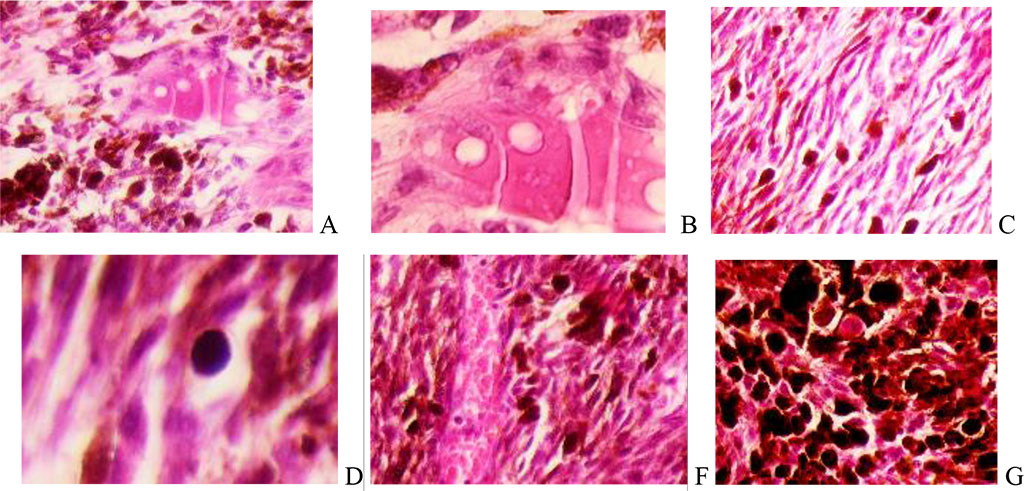
Figure 2. Melanoma of the choroid and ciliary body of the human eye. A, B - Epithelioid cells surrounded by melanocytes. C-spindle tumor cells. F and G – containing melanin. Hematoxylin and eosin staining. Magnification A, C, F, G x200; B, D x400.
In the preserved part of the ciliary body, the processes are characterized by the presence of foci of necrosis, degeneration of the pigment leaf and depigmentation of the vessels of the stroma of the processes of the ciliary body, as well as a large number of hypochromic cells, the absence of microvasculature vessels (Figure 3).
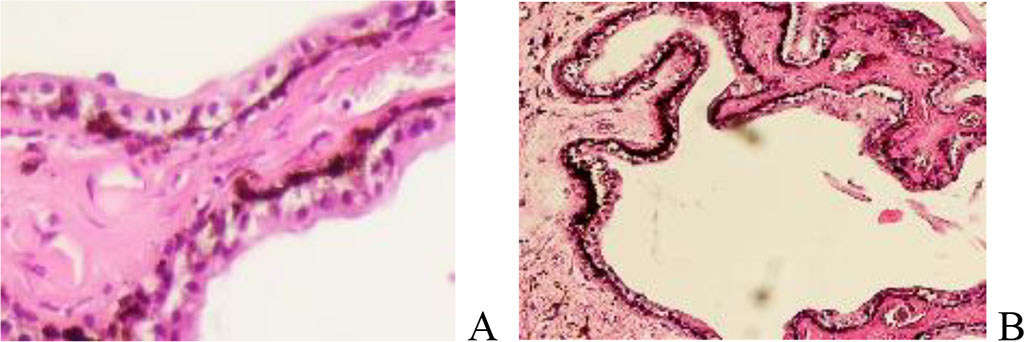
Figure 3. Ciliary process. Melanoma of the choroid and ciliary body of the human eye. Hematoxylin and eosin staining. Magnification A x400; B x100.
Using non-invasive methods as well as after enucleation, we found pathological forms of vessels on all sections, in some cases obliterated, sometimes with hypertrophied endothelium or similar to immature capillaries, which is consistent with the data of other authors. Identification and quantification of D2-40-positive lymphatic vessels in the ciliary body Khan A.M., Kagan D.B., Gupta N., et al. showed the presence of D2-40-positive lymphatic vessels exclusively in the peritumoral ciliary body. However, the authors noted that the lymphatic signal was significantly increased in the peritumoral ciliary body compared with the ciliary body in the malignancy zone (P < 0.0001) [7]. Thus, lymphatic vessels were not detected directly in the tumors, which served as the basis for the recommendation not to use this indicator in predicting metastasis, in contrast to blood vessels, the development of which, according to Folberg R., Mehaffey M., Gardner L.M., (1998), reflects invasive phenotype of tumor cells [8]. Using electron microscopy and immunohistochemical analysis, Bgatova N.P., Makarova V.V., Taskaeva Y.S., (2019) showed typical blood capillaries and vessels in uveal melanoma and identified various types of stromal cells in the tumor structure. Macrophages, fibroblasts of varying degrees of differentiation, and endothelial-like cells with numerous caveolae in the cytoplasm were found in the channels of the extracellular matrix surrounding accumulations of tumor cells. The presence of local structures in the extracellular matrix channels that stain positively for markers of blood and lymphatic vessels (CD31 and podoplanin) suggests that the described endothelial-like cells may be the structural basis of the blood and lymphatic vessels of the tumor [9]. Metastasis of uveal melanoma involves not only a developed native vascular network, but also developing tumor-retinal shunts and the identified new type of microcirculation in the CM with a high degree of structure of cell-free microcirculatory channels, independent of angiogenesis and consisting of an extracellular matrix surrounded by rows of tumor cells (“vasculogenic mimicry "). These channels are believed to play a major role in the metastasis of uveal melanoma [10]. Radiation exposure to the tissue surrounding the tumor is one of the reasons for subsequent long-term drug treatment against the background of decreased patient comfort and the need for inevitable enucleation of the eye. Radiation complications and lack of tumor regression lead to inevitable enucleation in the long term with a worsening vital prognosis; according to the Kaplan-Meier analysis, patients with uveal metastases show a 30% survival rate after 3 years and 24% after 5 years. The actual median survival of patients is low, from 3 to five years, since in most cases there is a failure of local treatment of large-sized tumors due to the high cellular activity of the remaining tumor cells and damage to the surrounding tissues [11, 12]. Secondary enucleation rates range from 5.8% during the first 12 months to 35–46.7% after 8 years and earlier [13, 14, 15]. To fully assess the pathogenetic changes in the developmental conditions in the structure of the choroid of melanoma, it is necessary to note the complete detachment of the retina, including its blind part (Figure 4).
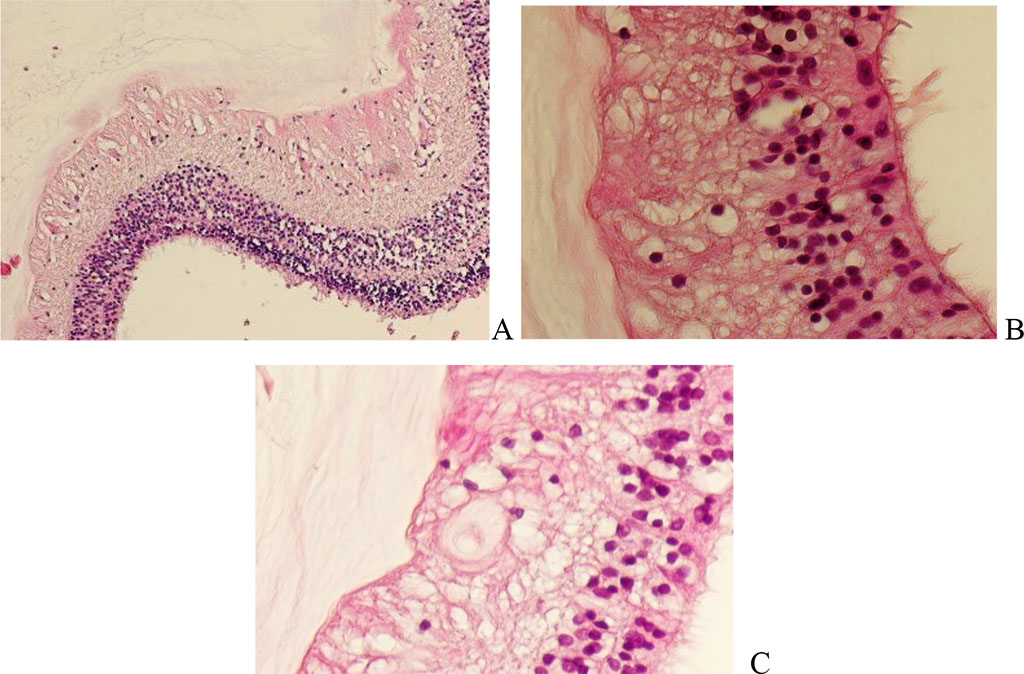
Figure 4. Detachment of the human retina against the background of melanoma of the choroid and ciliary body of the human eye. Hematoxylin and eosin staining. Magnification A x100; B x200; Сх400.
Retinal vessels with destroyed endothelium, hypertrophied chromophobe cells and destruction of retinal layers are identified. Shields C.L., Kalafatis N.E., Gad M., et al (2023) note the damaging role of metastases in the ocular structures surrounding the tumor [16]. Gündüz K., Shields J.A., Shields C.L., Eagle R.C. Jr. (1998) noted morphological and physiological pathological changes in Müller cells in the retina in uveal melanoma [17].
The results of hematoxylin and eosin staining of choroidal melanoma showed a tumor with centrifugal extension, without invasion of adjacent membranes and without ulceration. The obtained data on the morphological features of the structures surrounding the tumor expand the understanding of the morphology of uveal melanomas and can be used to develop personalized therapeutic targeted strategies. Morphological abnormalities in the structures surrounding melanoma indicate in favor of invasive treatment, since HD or local extirpation of melanoma will not improve the prognosis for the preservation of visual functions, but will worsen the vital prognosis in the long term. Lack of a favorable prognosis with a positive result of treatment, both at an early stage of tumor growth [18], with small sizes of CM against the background of a good primary therapeutic effect, the available world data on the risk of loss of visual functions with all modern methods of local treatment of CM [19, 20], as well as cases of rapidly growing tumor relapse 5 years or earlier after SRS, indicate in favor early enucleation as the gold standard for the treatment of uveal melanoma. Considering the fact of absence of benign melanomas, metastasis of intraocular melanomas at different times, inability to preserve visual functions and the presence of damage in all structures of the eye, regardless of tumor invasion in them, the treatment paradigm is to improve the vital prognosis.