- Home
- About the Journal
- Peer Review
- Editorial Board
- For Authors
- Reviewer Recognition
- Archive
- Contact
- Impressum
- EWG e.V.
BACKGROUND: Antibiotics have helped people to treat many diseases and have made the treatment of patients much easier. However, due to the development of resistance factors, antibiotics have become less effective against bacteria. Antimicrobial resistance is one of today's most important issues in global medicine.
OBJECTIVE: to determine the expression of cephalosporin drug resistance in an inpatient setting.
METHODS: This study was conducted at the Clinical Hospital of Emergency Medical Care Tver, Russia, for the period 2018-2020. Using the standard method of bacteriological examination, the microbiome of 624 patients was examined. The next step was to determine the level of antibiotic resistance.
RESULTS: Microorganisms of different spectrum, including Pseudomonas aeruginosa, Escherichia coli, Acinetobacter, etc., were isolated from clinical samples during the study. All microorganism groups showed decreased susceptibility to the drugs used in the study. Ceftriaxone (a third-generation cephalosporin) had the greatest loss of potency (reduction to 0% for all isolates in the study). Cefoperazone sulbactam and ceftazidime rapidly lost activity in this study. Increasing resistance to cefepime (a fourth-generation cephalosporin) was observed.
CONCLUSIONS. The study results indicate the rapid spread of resistance among a wide range of pathogenic microorganisms to the studied cephalosporin groups. Complete resistance developed within three years to the third-generation cephalosporins. A decrease in the susceptibility to the fourth generation of cephalosporins was observed.
KEYWORDS: cephalosporins, cefepime, ceftriaxone, antibacterial resistance.
The growth of bacterial resistance to antimicrobial drugs (AMR) becomes a serious problem in human life. The growing resistance of bacteria performs some modification in the treatment algorithms. The bacterial elimination is becoming more and more difficult for the health care systems worldwide. The antibiotic resistance control strategy remains the antibiotic prescription monitoring, the development of new drug groups. First AMR studies were published by scholars from different countries meanwhile the 3rd cephalosporin generation was prescribing widely. The increasing level of plasmid and chromosomal β-lactamase production limits the cephalosporin group to clinical practice.
During the last decades the five generations of cephalosporins have already been approved for clinical use. In spite of the significant amount the antibiotic's groups, the AMR is still a problem for the medical community. The present study demonstrates the AMR problem severity in a short (3-years) period inpatient setting in Tver, Russia.
The aim of this study was to determine the expression of cephalosporin drug resistance in an inpatient setting.
The standard method of bacteriological examination was used to study the microbiome in 624 inpatients aged from 18 to 95 years admitted to Clinical Hospital of Emergency Medical Care Tver, Russia in 2018-2020. In current study materials for bacteriological examination were wound discharge, pharynx and nose mucosal, cervical canal and vagina discharge, skin scrapings, urine, blood, sputum and other abdominal fluids.
The bacteriological examination consisted of obtaining material, transporting and seeding samples on nutrient media, incubation in an electric dry-air thermostat at 37°C. The period of bacterial exposure was 24 h.
To determine the level of antibiotic resistance we used paper discs saturated with Ceftriaxone, Cefepime, Cefoperazone+sulbactam and Ceftazidime. Levels of resistance of isolated microorganisms were assessed by estimating the diameter of the stop zone around the testing drugs discs.
The cephalosporin group is the most prevalent antibiotic drug used to treat infections at the Clinical Hospital of Emergency Medical Care Tver, Russia. This group is prescribed widely due to its high bactericidal activity, low toxicity and absence of evident antagonism with other drugs.
The increasing prescription of antibacterial drugs develops the multiple bacteria resistance. The β-lactamase production is one of the most important mechanisms of cephalosporin's inhibition.
The medical community needs to control the widespread use of cephalosporins [1].
This study demonstrated the regression of the cephalosporin drug’s bactericidal effect.
Escherichia coli showed high antibiotic resistance to a wide range of drugs, including cephalosporin group (Tab.1, fig. 1). Third- and fourth-generation Cephalosporins were mainly being developed for the treatment patients infected with Enterobacteriaceae. The efficacy of Ceftriaxone in eliminating E. coli in 2018 was 72.6%. This value decreased to 5.8% in 2019, reached zero, in 2020 hence the drug became totally ineffective. Cefepim showed high efficacy - 60% in 2018. The high efficacy of the drug in the treatment of Escherichia coli was demonstrated later 40% in 2019 and 25% in 2020.
Staphylococcus aureus initially showed high resistance to cephalosporin drugs (Tab.1, fig.2). 15% of Staphylococcus aureus were sensitive to Ceftriaxone in 2018. The eliminating efficacy of Ceftriaxone decreased to 4.5% in 2019 and reached 0% in 2020. As a result of Ceftriaxone prescription is the absolute drug resistance in an inpatient setting. The Staphylococcus aureus sensitivity to Cefepime was 10% in 2018. Subsequently, the sensitivity increased to18% in 2019 and almost returned to the initial value (8.33%) in 2020. A literature search showed the similar data of the Staphylococcus aureus sensitivity [2, 6].
According to the current study results almost half of the strains of Klebsiella pneumoniae were sensitive Ceftriaxone in 2018 (45.5%). Klebsiella pneumoniae demonstrated the total resistance to Ceftriaxone in 2019 and 2020. Cefepime provided low antimicrobial effect - 18% in 2018, 11.9% in 2019 and 10.5% in 2020 (Tab.1, fig.3). Gram-negative Klebsiella oxytoca showed high sensitivity to Cefepime, a 4th generation cephalosporin (Tab.1). Cefepime eliminated 80% of Klebsiella oxytoca in 2018, 17% in 2019, and later the sensitivity level reached 50% in 2020.
Gram-negative Acinetobacter strains were mainly found in the genitourinary tract and in the discharge from wounds. Further investigation showed the high level of cephalosporin resistance during 2018-2022 (Table 1, Figure 4). According to the study, ceftriaxone neutralised half of the strains (50%) in 2018, 27.27% in 2019 and zero sensitivity in 2020. Cefepime sensitivity was 66.67% in 2018. Total cefepime resistance was detected in 2019 and 2020.
Pseudomonas aeruginosa showed a high level of sensitivity to the cephalosporins. In the first year of the study, the efficacy of the ceftriaxone used against Pseudomonas aeruginosa was high (71.4% of the strains were eliminated), but then this value decreased first to 18.5% in 2019 and then to 0 in 2020. Cefepim showed a satisfactory rate of elimination of Pseudomonas aeruginosa over the whole study period (57 % in 2018, 15 % in 2019 and 33 % in 2020).
The combination with sulbactam was completely ineffective throughout the study. The bacteria also developed complete resistance to ceftazidime in this study. However, they are widely used due to their convenience [1, 5, 9].
The data presented in this study primarily demonstrate the prevalence of β-lactamases in a wide range of microbial strains. The high level of resistance to cefepime, ceftazidime, cefoperazone-sulbactam and ceftriaxone in most isolates indicates the widespread distribution of extended-spectrum β-lactamases in the sample of strains in this study. This often leads to complications in treating patients when this group of drugs is used. In clinical practice, the almost complete absence of positive dynamics can sometimes be observed.
Our findings were often consistent with other studies on antimicrobial resistance. However, some of them provided new data on the topic, the individual principles of therapy in different treatment and prevention settings, and the impact of the health care system on knowledge, attitudes and patterns of antibiotic use [3, 4, 7, 8].
Conclusion. The present study shows an accelerated rate of resistance development to cephalosporins in a short period of time in an outpatient setting. The relatively high level of losses indicates an urgent need to review current antibiotic therapy practices. Strategies to combat antimicrobial resistance must be provided by the medical community. We need to monitor prescribing, optimise antimicrobial use, and educate patients and healthcare providers about the problem and ways to control the AMR worldwide.
Table 1
| Antibiotic | Isolates | Year | |||||||||||||||
| Acinetobacter | Staphylococcus aureus | Klebsiella pneumoniae | Pseudomonas aeruginosa | Staphylococcus saprophyticus | Escherichia coli | Klebsiella oxytoca | Staphylococcus epidermidis | ||||||||||
| 1* | 2* | 1* | 2* | 1* | 2* | 1* | 2* | 1* | 2* | 1* | 2* | 1* | 2* | 1* | 2* | ||
| % | n | % | n | % | n | % | n | % | n | % | n | % | n | % | n | ||
| Ceftriaxone | 50 | 6 | 15 | 20 | 45,45 | 11 | 71,43 | 7 | - | 8 | 72,6 | 69 | - | 5 | 0 | 9 | 2018 |
| 27,27 | 11 | 4,55 | 22 | 0 | 42 | 18,51 | 27 | - | 16 | 5,77 | 49 | - | 6 | 0 | 12 | 2019 | |
| 0 | 7 | 0 | 12 | 0 | 19 | 0 | 24 | - | 12 | 0 | 57 | - | 6 | 0 | 14 | 2020 | |
| Cefepime | 66,7 | 6 | 10 | 20 | 18 | 11 | 57,14 | 7 | 0 | 8 | 60.27 | 69 | 80 | 5 | 33,3 | 9 | 2018 |
| 0 | 11 | 18,2 | 22 | 11,9 | 42 | 14,81 | 27 | 6,25 | 16 | 38,46 | 49 | 16,7 | 6 | 8,3 | 12 | 2019 | |
| 0 | 7 | 8,33 | 12 | 10,53 | 19 | 33,33 | 24 | 0 | 12 | 25 | 57 | 50 | 6 | 0 | 14 | 2020 | |
| Cefoperazone-sulbactam | 0 | 6 | - | 20 | 0 | 11 | - | 7 | - | 8 | 0 | 69 | - | 5 | - | 9 | 2018 |
| 0 | 11 | - | 22 | 0 | 42 | - | 27 | - | 16 | 0 | 49 | - | 6 | - | 12 | 2019 | |
| 0 | 7 | - | 12 | 11 | 19 | - | 24 | - | 12 | 0 | 57 | - | 6 | - | 14 | 2020 | |
| Ceftazidime | - | 6 | 0 | 20 | - | 11 | - | 7 | - | 8 | - | 69 | - | 5 | - | 9 | 2018 |
| - | 11 | 9 | 22 | - | 42 | - | 27 | - | 16 | - | 49 | - | 6 | - | 12 | 2019 | |
| - | 7 | 0 | 12 | - | 19 | - | 24 | - | 12 | - | 57 | - | 6 | - | 14 | 2020 | |
1*: percentage of
strains sensitive to the drug.
2*: number of
pathogens isolated.
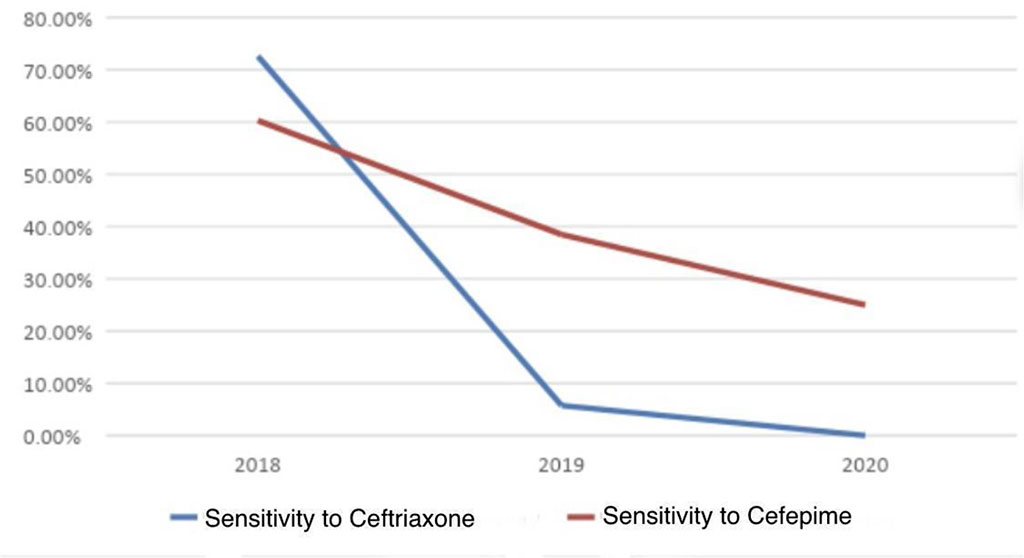
Figure 1: Escherichia coli
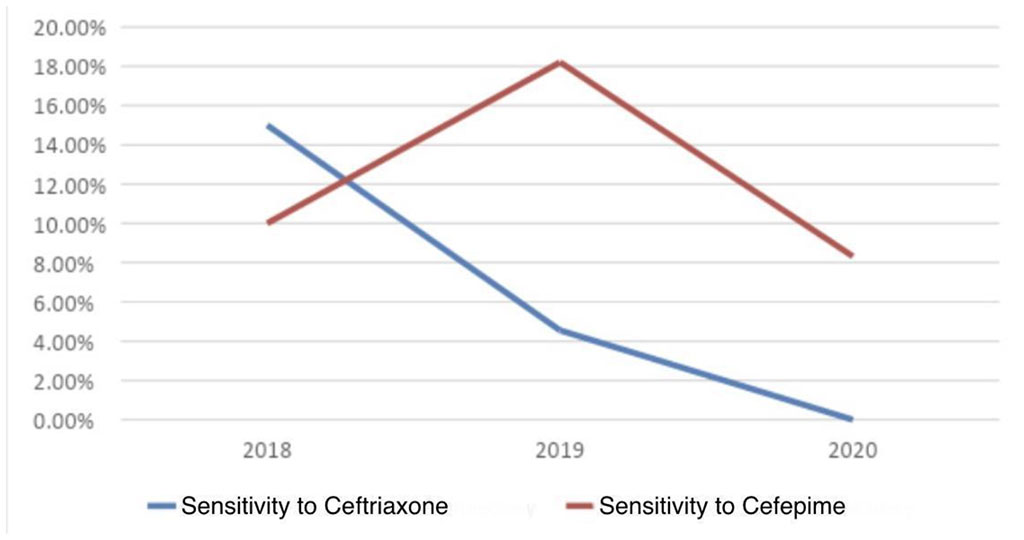
Figure 2: Staphylococcus aureus
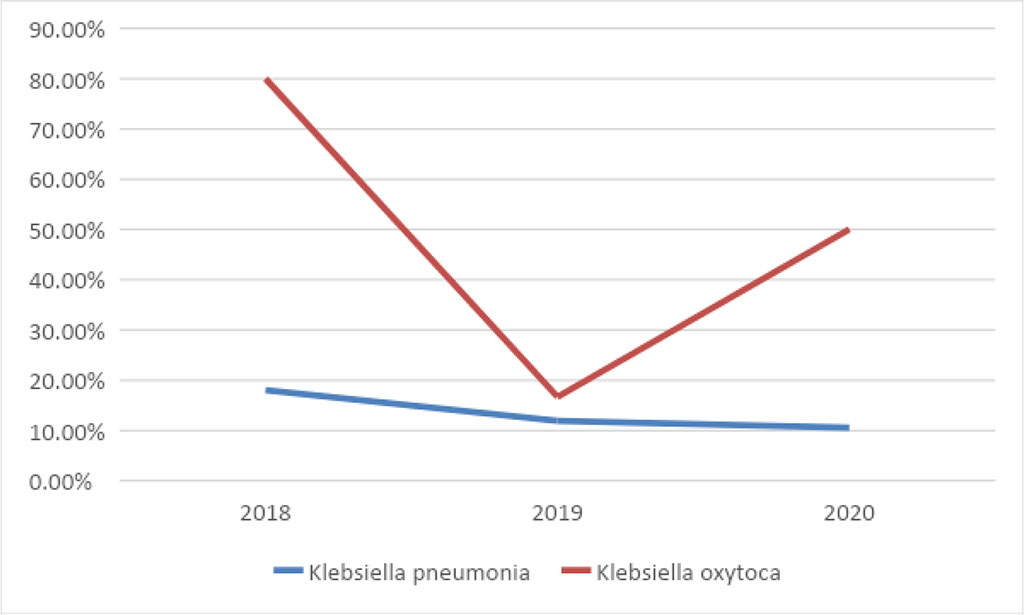
Figure 3: Klebsiella pneumoniae
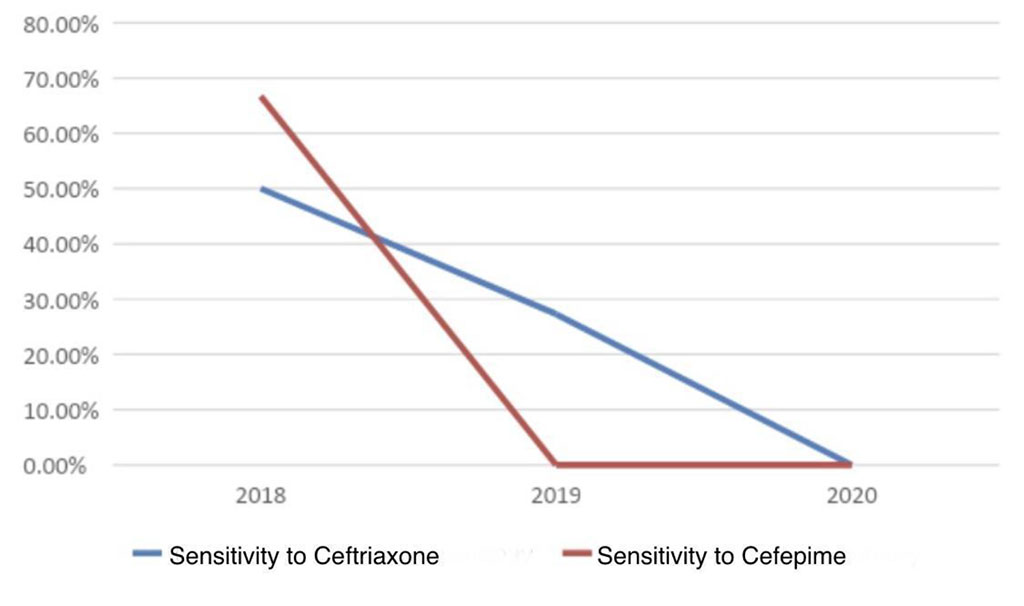
Figure 4: Acinetobacter