- Home
- About the Journal
- Peer Review
- Editorial Board
- For Authors
- Reviewer Recognition
- Archive
- Contact
- Impressum
- EWG e.V.
Red bone marrow samples investigation in deceased COVID-19 patients enabled to identify the phenomena of secondary hemophagocytosis. Analysis of the data showed that phagocytic reactions during infection of patients with SARS-CoV-2 are manifested both in relation to erythrocytes and leukocytes. The data obtained make it possible to expand the strategy of therapeutic measures, taking into account the new data on the mechanisms of the pathogenesis of COVID-19 in severe viral infection based on morphological findings and additional information on the involvement of young erythrocytes and lymphocytes in the structure of the red bone marrow in the cascade of pathological reactions. The results obtained confirm a wide range of aggressive damaging effects of SARS-CoV-2 in the development of multiple organ failure against the background of COVID-19 and the involvement of the red bone marrow in the pathological process. The authors supplemented information about the mechanisms of hypoxia in COVID-19, which is not only a consequence of damage to the respiratory epithelium, but also the result of damage to erythrocyte differons both at the level of red bone marrow and in peripheral blood. This fact must be taken into account in the development of a treatment strategy and in the creation of new drugs for the treatment of infected patients with various strains of SARS-CoV-2.
Keywords: secondary hemophagocytosis, erythrocytes, leukocytes, ischemia, hypoxia, hyperinflammation, cytokine storm, red bone marrow, hematopoiesis.
Relevance Despite optimistic forecasts for improving the epidemiological situation associated with the COVID-19 pandemic, mortality and post-COVID complications remain at a high level [24], and the mechanisms of the pathogenesis of multiple organ failure require further research against the backdrop of the emergence of new strains of SARS-CoV-2 [1, 5, 20]. Hyperinflammation against the background of COVID-19 with uncontrolled proliferation of immunocytes / phagocytes, caused by viral induction of activation of lymphocytes and macrophages, against the background of their morphological goodness, is accompanied by a cytokine storm [14, 16, 19] and secondary hemophagocytic lymphohistiocytosis (HLH), which is a reflection of a life-threatening and prognostic condition [2, 4, 9]. It is known that secondary hemophagocytic lymphohistiocytosis (HLH) in clinical practice in most cases occurs in cancer patients. [25], due to aging [22], diagnosed by triglyceride levels, low fibrinogen levels, transaminitis and elevated ferritin levels, the iron transfer mediator transferrin, and D-dimer levels [8, 13, 17]. These indicators are only indirect confirmation of the presence of phagocytic reactions in peripheral blood and red bone marrow (RMB), based on previously obtained data on HLH associated with mononucleosis and Epstein-Barr virus. [3, 11, 15, 27]. However, in COVID-19, mechanisms leading to anemia and iron deficiency symptoms based on reduced ferritin levels [7] and their diagnostic significance against the background of unstable indicators of erythropenia, leukocytopenia and thrombocytopenia at the present stage are not known. The association of SARS-CoV-2 infection with HLH is little studied. [21, 23, 26] This determined the direction of our research.
The aim of the study was to identify the morphological parameters of red bone marrow (RMB) damage during SARS-CoV-2 infection.
The study was conducted in accordance with the Declaration of Helsinki 2000 and its edition since 2013. The design and protocol of the study were approved by the Ethics Committee of the Far Eastern Federal University No. 4 dated March 22, 2021. The study used biopsy specimens obtained from 43 patients who died as a result of COVID-19 disease caused by SARS-CoV-2. Morphological studies were carried out using hematoxylin and eosin staining of decalcified sections of red bone marrow. The illustrative material was obtained using microscopic equipment from Olympus (Japan).
We have found that moderate to severe hemophagocytosis is observed in the red bone marrow of deceased patients with a PCR-confirmed study for the detection of SARS-CoV-2 viral infection etiology. At the same time, phagocytosed cells were identified in red bone marrow (RBM) macrophages, which are both precursors of the myeloid and lymphocytic germs. Analysis of the revealed differences in hemophagocytosis in RBM and in peripheral blood smears presented in the works of other authors [6, 12, 18]. Our studies have shown true phagocytosis, which is not associated with cell capture without disrupting the structure of the cell membrane and nucleus, but with the destruction of the cyto- and karyolemma, and the fusion of the cytoplasm. At the same time, not only erythrocytes, but also granulocytes and lymphocytes were identified in the hyaloplasm of macrophages. (Figure 1, 2).
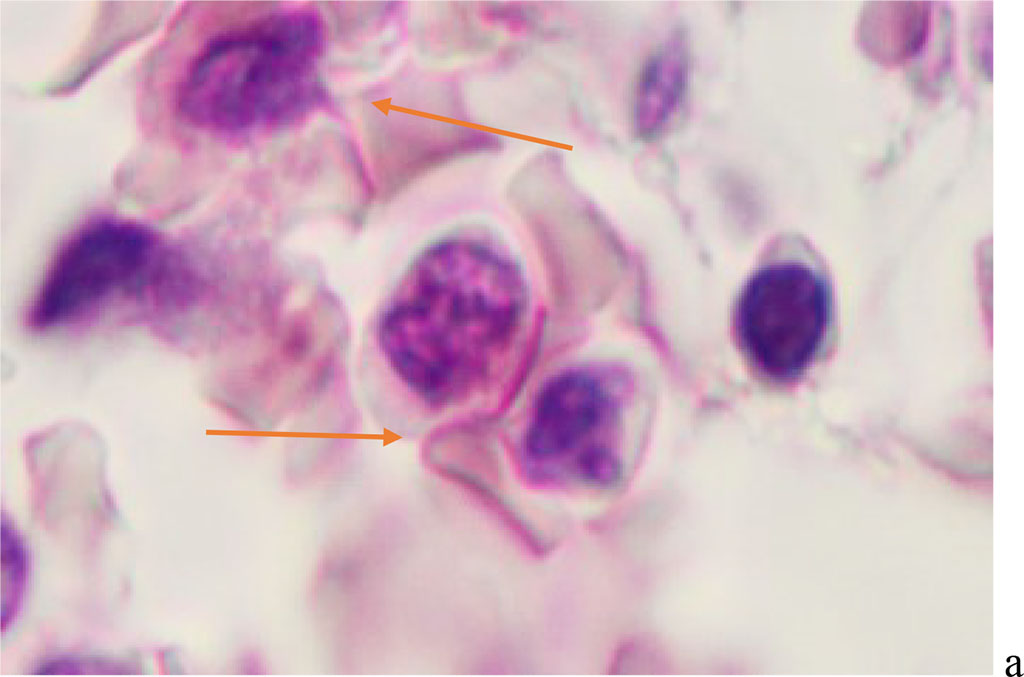
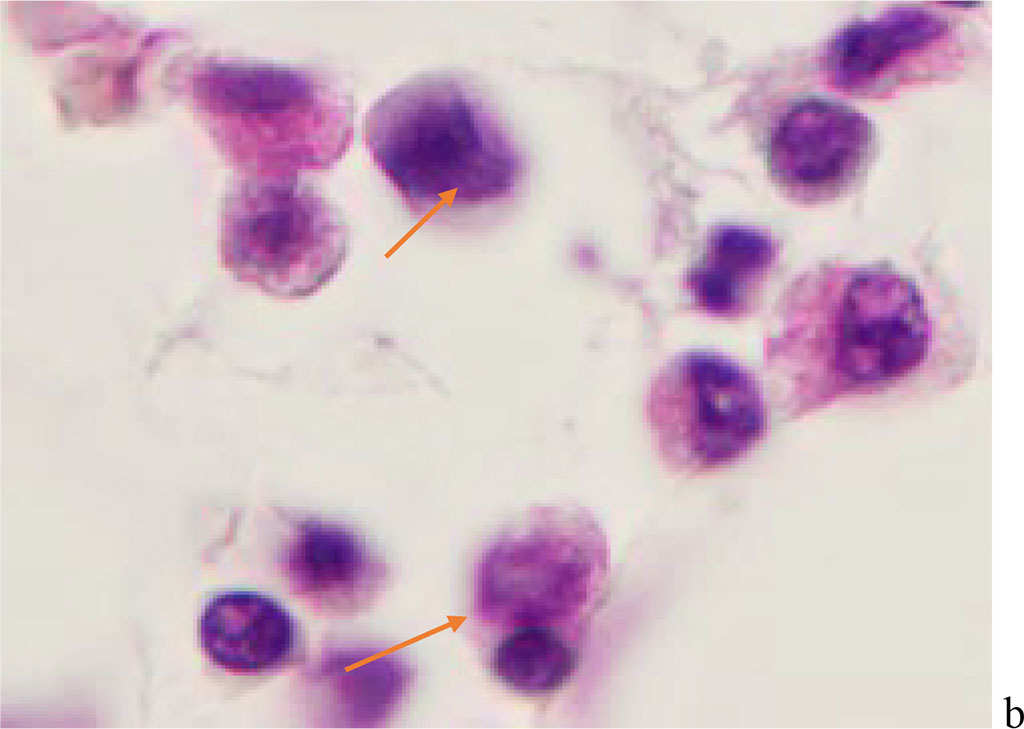
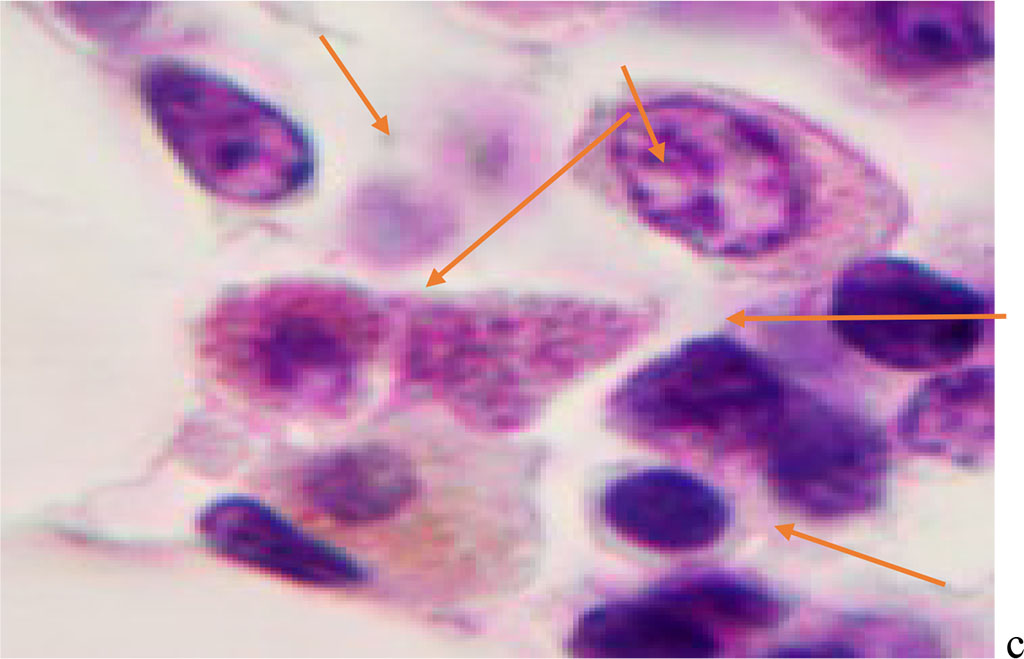
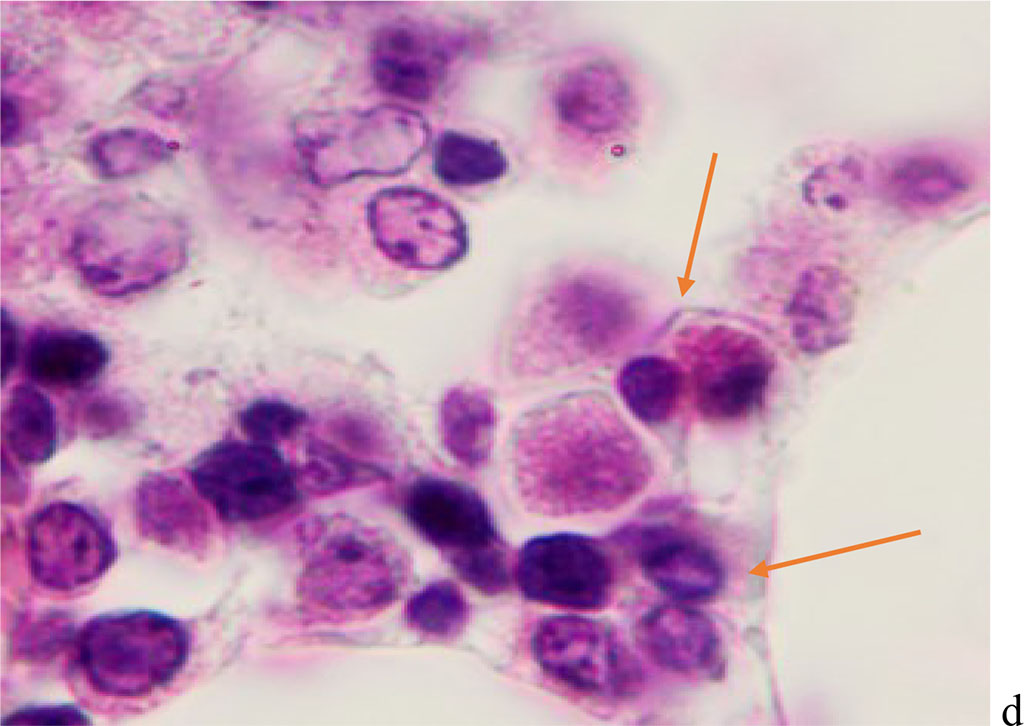
Figure 1 - Red bone marrow. Stained with hematoxylin and eosin. Magnification x 400.
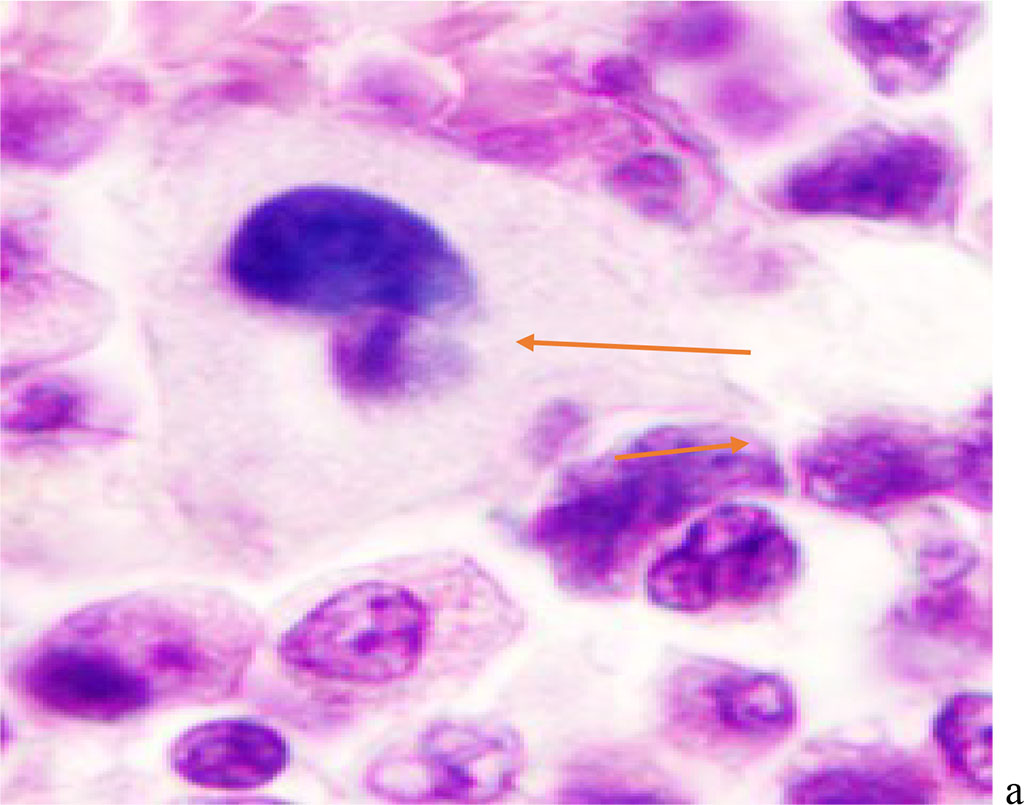
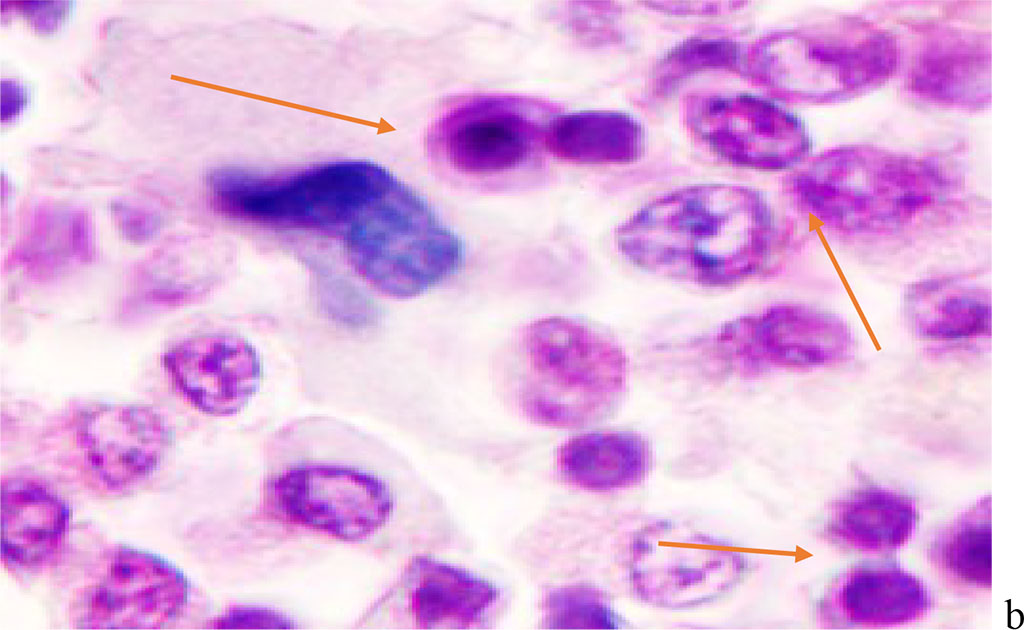
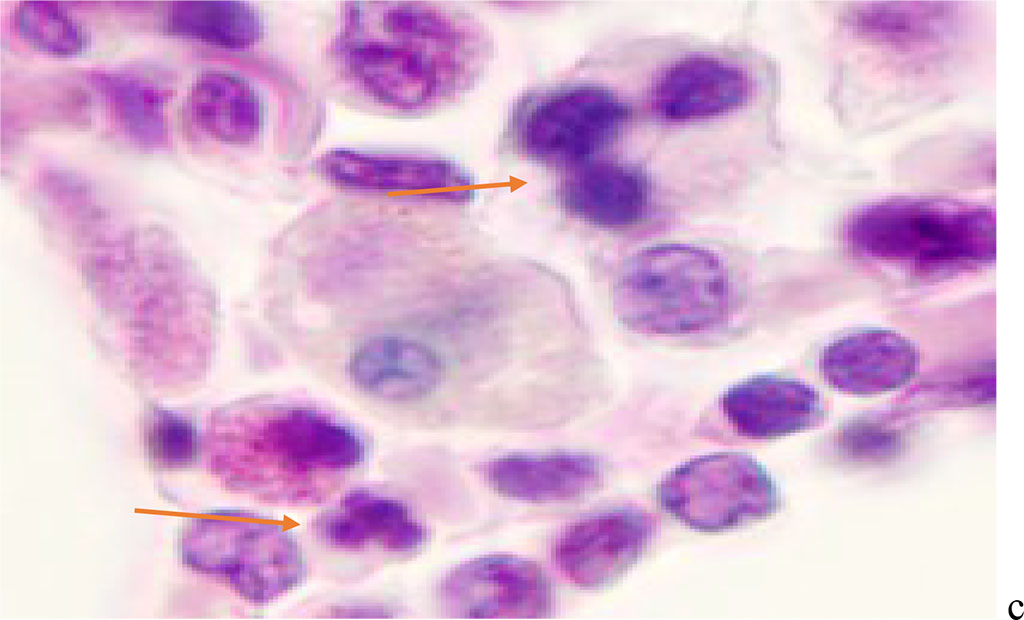
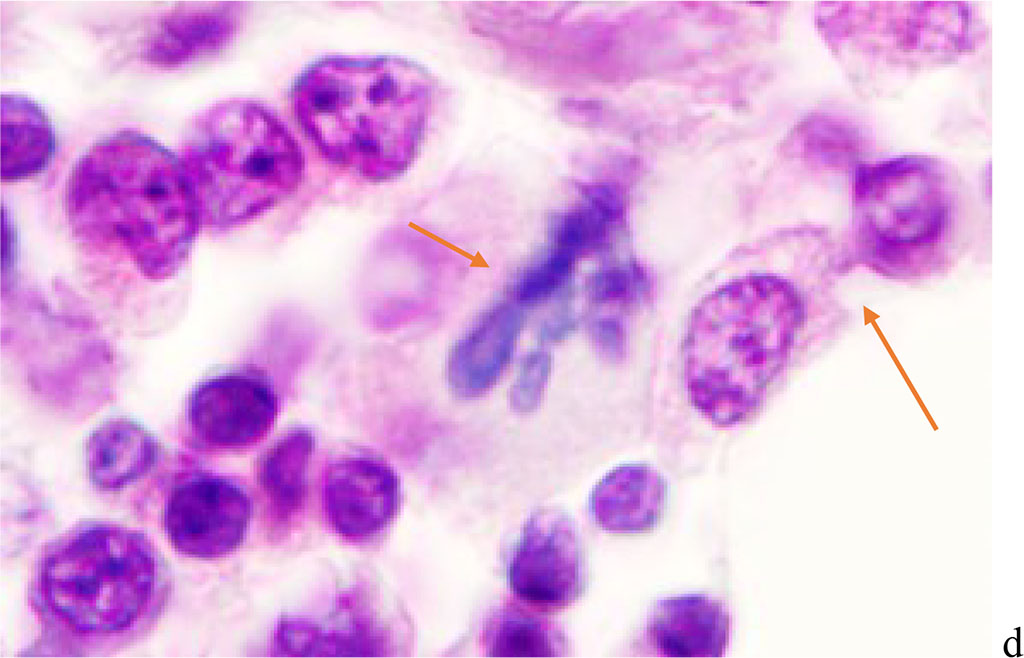
Figure 2 - Red bone marrow. Stained with hematoxylin and eosin. Magnification x 400.
We noted that hemophagocytosis in BMC during SARS-CoV-2 infection was characterized not only by the fusion of the cytoplasm, but also by apoptosis of the nuclei of phagocytosed cells, identified by the ring-shaped arrangement of heterochromatin in the nuclei. We noted that some cells contain several apoptotic nuclei (Figure 3, 4). We assume that the physiological significance of hemophagocytosis in BMC is not associated with the destruction of apoptotic cells, but is the result of a deficiency in the trophic supply of developing progenitor cells of myeloid and lymphoid germs, iron deficiency, and hypoxia.
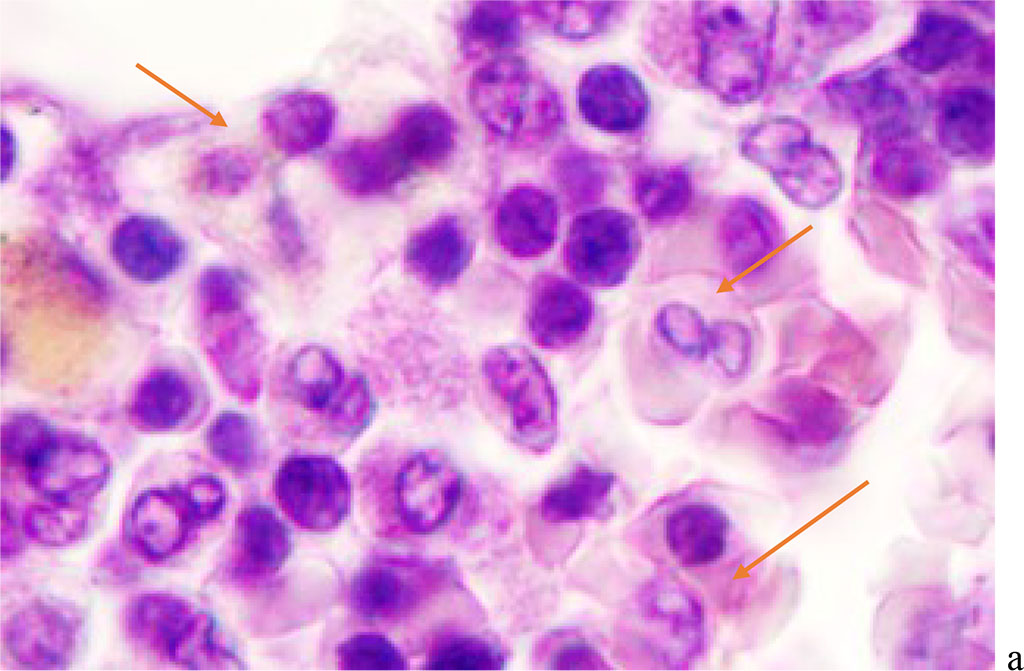
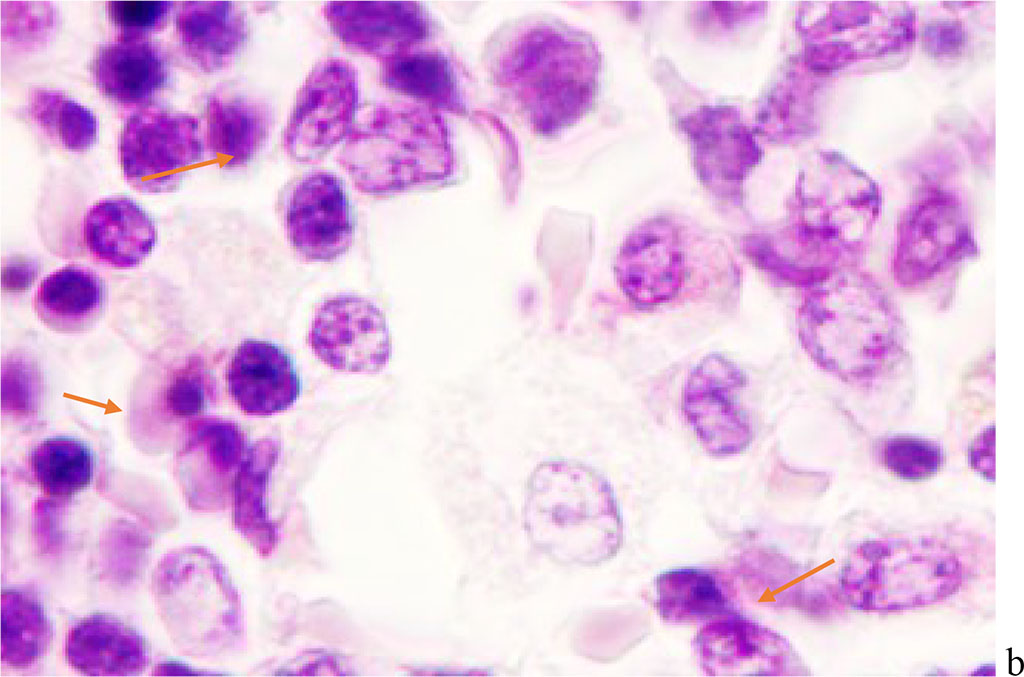
Figure 3 - Red bone marrow. Stained with hematoxylin and eosin. Magnification x 400.
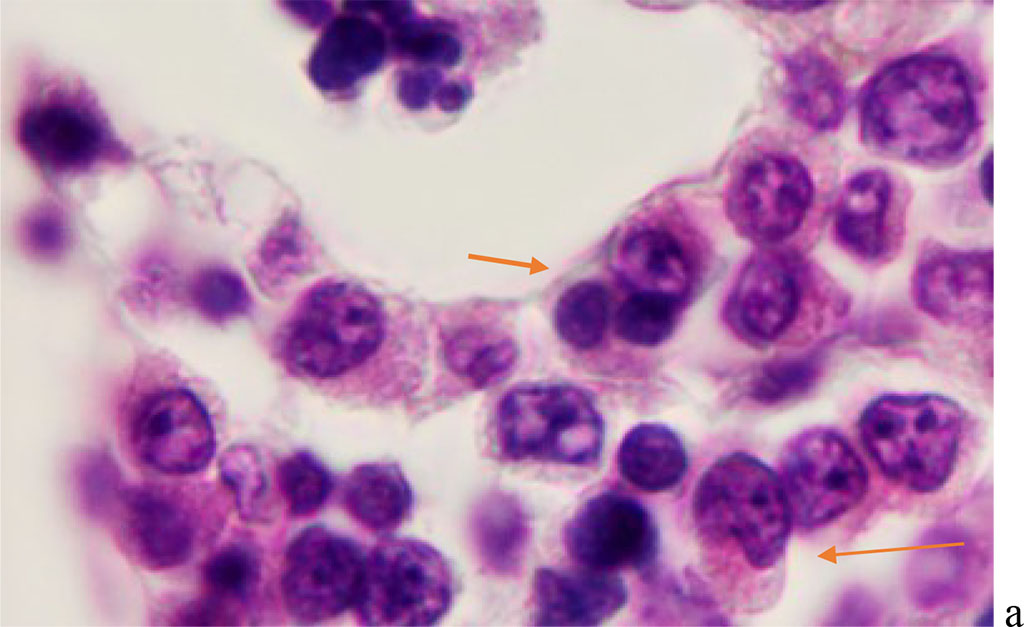
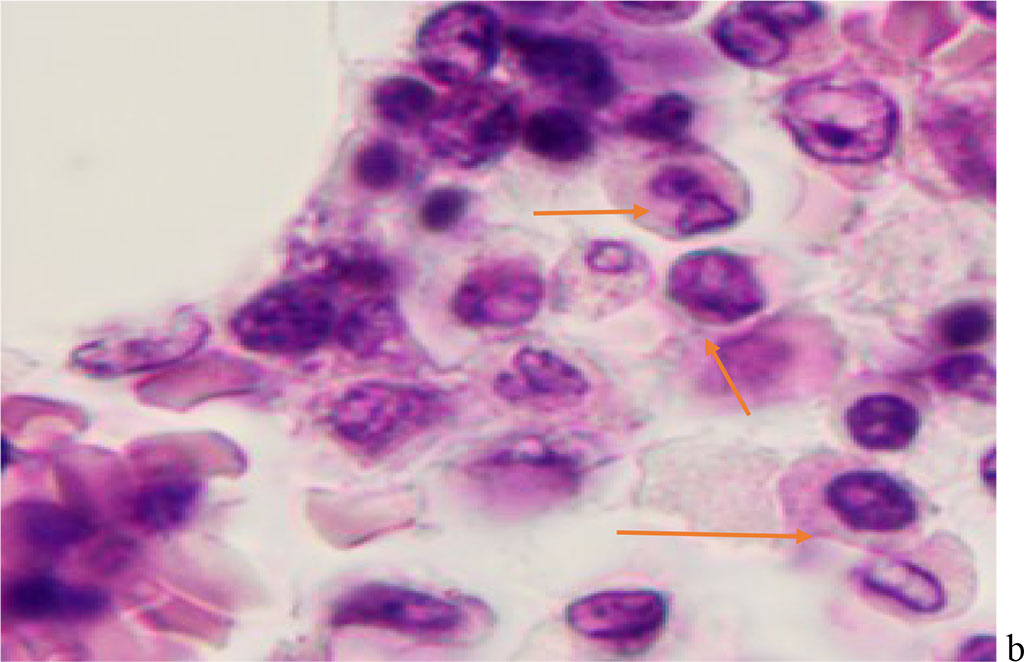
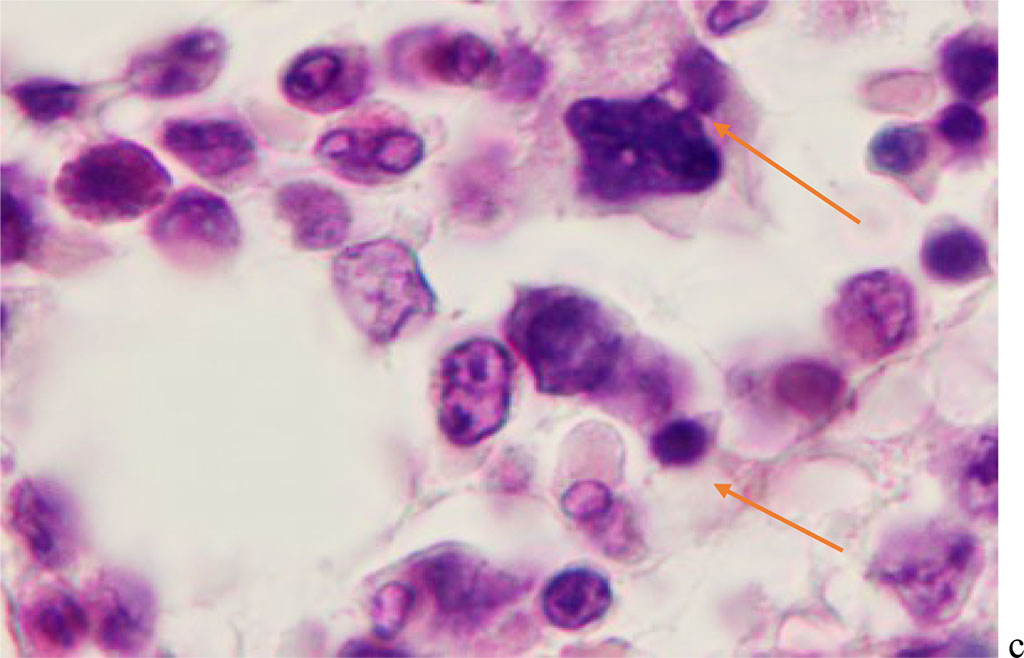
Figure 4 - Red bone marrow. Stained with hematoxylin and eosin. Magnification x 400.
Hemophagocytosis in BMC of patients with viral infection with SARS-CoV-2 etiology is characterized by the participation in this process not only of agranulocytic macrophages, but also of cells with eosinophilic granules in the cytoplasm. At the same time, the participation of eosinophilic granulocytes both in the phagocytosis of cells of other differons, and their destructive interactions with each other with the development of apoptosis in one of the eosinophilic cells (Figure 5).
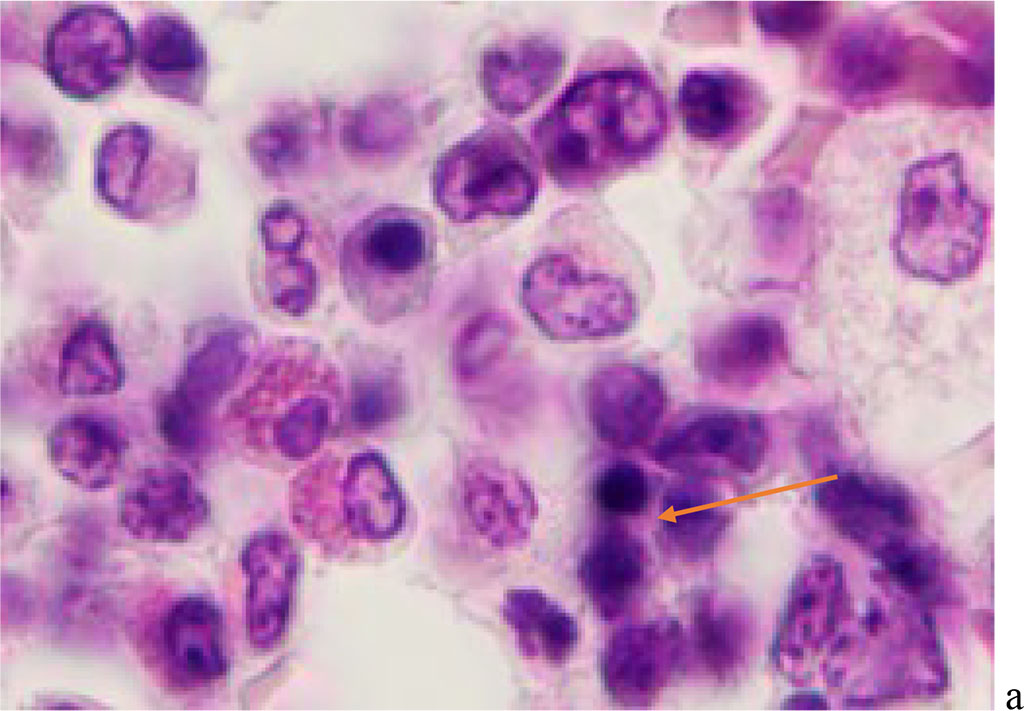
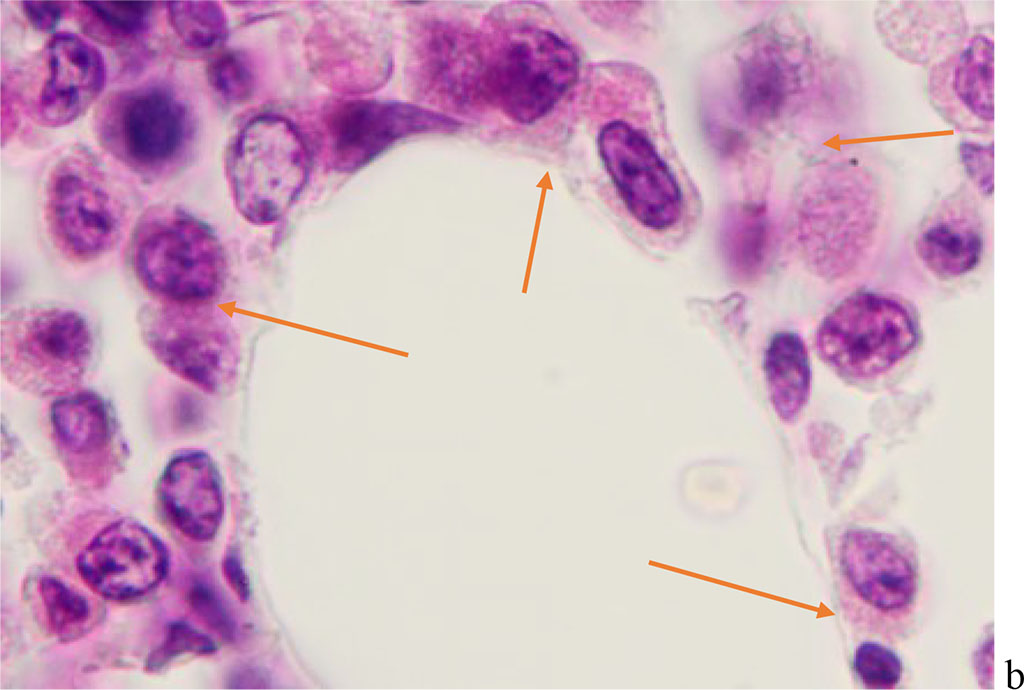
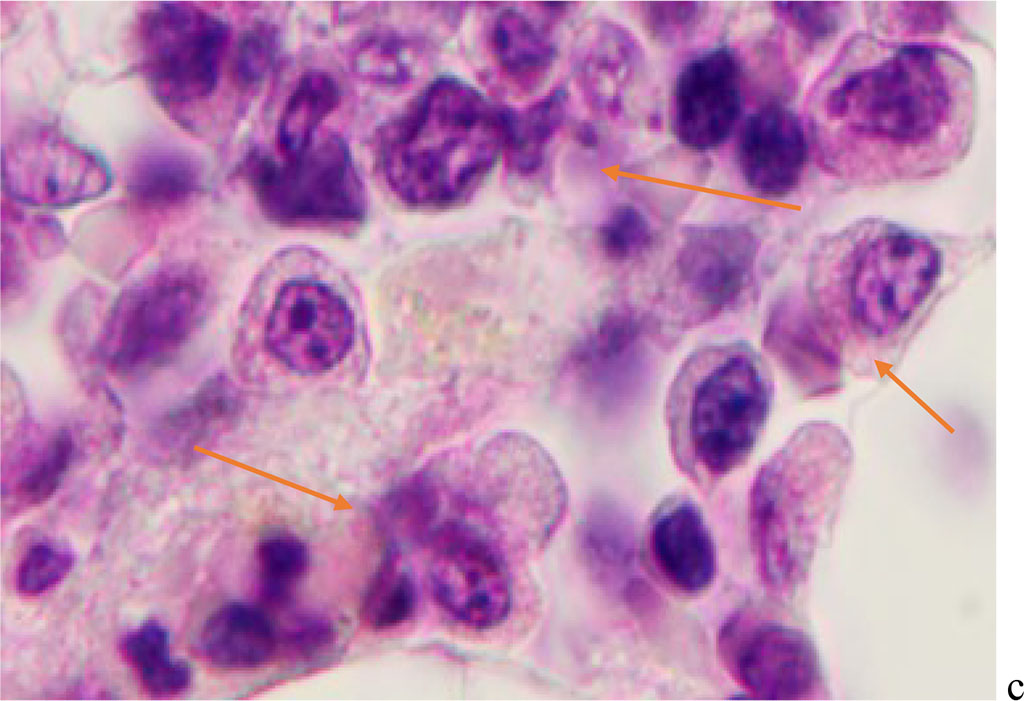
Figure 5 - Red bone marrow. Stained with hematoxylin and eosin. Magnification x 400.
Phagocytic cells have a structured nuclear membrane, a cellular cytoplasm at the light-optical level, uneven and fuzzy boundaries surrounded by progenitor cells with round or oval nuclei, and a narrow rim of basophilic cytoplasm (Figure 6).
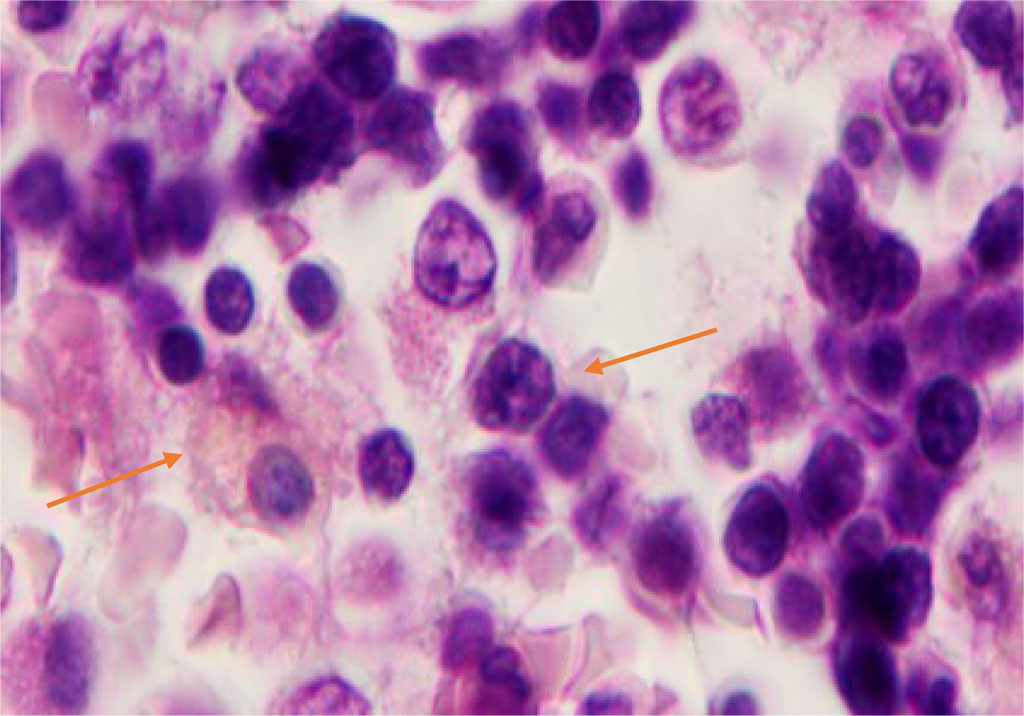
Figure 6 - Red bone marrow. Stained with hematoxylin and eosin. Magnification x 400.
On preparations of red bone marrow of this group of patients, 1-2 hypochromic, basophilic and oxyphilic erythrocytes were observed in the field of view, differing in size and irregular shape, and 1-2 macrophages with iron granules and phagocytosed erythrocytes were identified. Many coronaviruses are known to induce autophagy. Formerly H. Chandra, S. Chandra, Rm. Kaushik, Nk. Bhat, V. Shrivastava (2014) in bone marrow aspirates described the phenomena of hemophagocytosis, which is a life-threatening condition, based on the identification of macrophages with phagocytosed neutrophils and a large number of plasma cells against the background of pancytopenia and erythrocyte hyperplasia [6]. However, morphologically, in other viral infections, the studied cells of BMC aspirates are characterized by a membrane, cytoplasm, and nucleus that are practically unchanged during their phagocytosis, which may be associated with viral damage to the cell absorbed by the macrophage for its further isolation from the surrounding tissue. During SARS-CoV-2 infection, phagocytosis differs not only from the primary one by the absence of changes in the genome, but also from the secondary one in other viral infections, as it is associated with the adaptation of emerging hematopoietic sprouts to a lack of signaling molecules for inducing hematopoiesis and trophic supply, the absence of iron, and hypoxia. This phagocytosis is aimed at an attempt to provide a part of the emerging cells for their full differentiation and specialization. Therefore, in the morphological picture of BMC sections, many apoptotic and damaged cells with pronounced karyopyknosis are identified. The findings suggest that hemophagocytosis, even if seen in single cells, should always be documented in bone marrow diagnostic reports. Bone marrow aspirates should be included in the differential diagnosis of COVID-19 caused by SARS-CoV-2. This may be the only sign of the development of COVID-19 infection caused by SARS-CoV-2, with damage to the red bone marrow and predicting a severe course of COVID-19, as well as for the development of a pathogenetically sound strategy.
Monitoring and analysis of intercellular interactions under the influence of viral cellular damage is crucial for understanding the pathogenesis of the disease for the development of prevention and targeted treatment through the protection and regeneration of damaged targets. These studies are of particular importance given the global problem of COVID-19 caused by increasingly aggressive strains of SARS-CoV-2 [18].
Understanding the spectrum and frequency of histological findings in COVID-19 is essential for a better understanding of the mechanisms of pathogenetic aspects of the disease and the prospect of predicting favorable outcomes. Hemophagocytosis, observed in the red bone marrow of dead patients with COVID-19, must be taken into account in the protocols for the study of the material, since this is a marker of deep damage not only to the central organ of hematopoiesis, like red bone marrow, but also to systems involved in the regulation of hematopoiesis, leading to hypoxia and death. In conditions of iron deficiency, it is necessary to take into account the mechanisms of increasing the level of ferritin in the blood when prescribing iron preparations, since in case of a redistributive deficiency; they can lead to secondary hemosiderosis with subsequent aggravation of the patient's condition. Bone marrow aspirates should be included in the differential diagnosis of COVID-19 caused by SARS-CoV-2. This may be the only sign of the development of COVID-19 infection caused by SARS-CoV-2, with damage to the red bone marrow and with a prediction of severe course of COVID-19, as well as for the development of a pathogenetically sound strategy with the use of blood transfusion or BCM transplantation for life reasons of healthy donors. Thus, the pathogenesis of HLH in severe COVID-19 is associated not only with hypoxia in case of damage to the alveolar epithelium, but also due to damage to the stroma and blood vessels of the BCM, abnormal activity of lymphocytes and macrophages in the tissue of the red bone marrow. There are also disturbances in the hemoglobin synthesis regulation system and damage to erythrocyte differon and erythropenia, which leads to disruption of cellular respiration and severe damage to organs involved in the synthesis of erythropoietin.
PCR-confirmed diagnosis of infection with SARS-CoV-2 strains does not require the expectation of a high index of suspicion for HFH based on the proposed eight criteria for HFH, as a delay will lead to an increase in mortality.
Peripheral blood hemogram parameters and biochemical analyzes will be lagging in comparison with earlier pathological processes in the structure of the red bone marrow.
To predict and timely prevent death, it is necessary to study the aspirates of the BCM for immediate relief of damage to its structure.